FDA-Approved Treatment for Psoriasis
Betamethasone dipropionate spray, 0.05% (Sernivo)
This medication was approved by the FDA in February 2016 and is indicated for the treatment of mild to moderate plaque psoriasis in people ages 18 and older. It helps relieve symptoms like redness, flaking, and itching by suppressing the body’s inflammatory response.
Betamethasone dipropionate spray rapidly relieved bothersome symptoms of psoriasis and improved psoriatic signs in hard-to-treat knee and elbow plaques. [1]
In subjects with moderate plaque psoriasis, betamethasone dipropionate outperformed vehicle spray in the percentage of subjects with a reduction of at least 50 percent in the Total Sign Score (TSS), which is the sum of individual scores for erythema or redness, scaling, and plaque elevation. [2]
In subjects with moderate plaque psoriasis, betamethasone dipropionate spray 0.05% resulted in good levels of patient satisfaction and quality of life measures as evidenced by improvements in the global assessment of disease and the level of itching experienced by subjects. [3]
Halobetasol propionate-tazarotene lotion, 0.01%/0.045% (Duobrii)
Approved by the FDA in April 2019 for the treatment of plaque psoriasis in adults, this medication is a combination of a corticosteroid (halobetasol propionate) and a retinoid (tazarotene). It works by suppressing inflammation and inhibiting the growth of excess skin cells.
In patients with plaque psoriasis, the application of Duobrii lotion for 1 year was associated with a significant reduction in symptoms in 58.8% of participants. [4]
In non-White, White, and Hispanic/Latino participants with psoriasis, Duobrii treatment resulted in numerically greater reductions in affected body surface area (BSA). [5]
Halobetasol propionate foam, 0.05% (Lexette)
This topical corticosteroid was approved by the FDA for the treatment of plaque psoriasis in May 2018 as a generic and it became available under the brand name Lexette in April 2019.
In adolescents aged 12 to 17 years old, the use of hyalobetasol propionate foam was associated with a tolerable safety profile. [6]
Halobetasol propionate lotion, 0.01% (Bryhali)
Halobetasol propionate lotion, 0.01 percent (Bryhali) got its FDA approval in November 2018. This is indicated for the treatment of plaque psoriasis in adults.
Once daily application of halobetasol propionate lotion 0.01% for 8 weeks in patients with plaque psoriasis improved the symptoms with mild to moderate adverse effects. [7]
In patients with moderate to severe plaque psoriasis, once daily application of halobetasol propionate lotion 0.01% for 8 weeks resulted in improvement in psoriasis signs and symptoms and body surface area. [8]
In male and female participants with moderate-to-severe psoriasis, daily application of halobetasol propionate lotion 0.01% for 8 weeks was associated with significant reductions in disease severity. [9]
Certolizumab pegol (Cimzia)
This medication was approved by the FDA for the treatment of moderate to severe plaque psoriasis in May 2018. Cimzia is recommended for patients who are candidates for phototherapy or systemic therapy. It reduces inflammation by blocking TNF-alpha.
In patients with chronic plaque psoriasis, treatment with either certolizumab 400 mg or 200 mg every 2 weeks was associated with significant and clinically meaningful improvements in the signs and symptoms of the disease. [10]
In patients with moderate to severe psoriasis, certolizumab produced significant improvement in symptoms after 3 months of treatment and was maintained for the 12-month analysis period. [11]
In patients with chronic plaque psoriasis, both certolizumab regimens improved psoriasis symptoms, with a greater response observed with the higher dose. [12]
Tildrakizumab-asmn (Ilumya)
This medication was FDA-approved in March 2018 for the treatment of plaque psoriasis in adults who are potential candidates for other treatments such as phototherapy or systemic therapy. Tildrakizumab-asmn suppresses inflammation by blocking IL-23.
In patients with moderate to severe plaque psoriasis, tildrakizumab treatment improved health-related quality of life with higher tolerability, both in the short- and longer-term. [13]
In patients with moderate to severe chronic plaque psoriasis, tildrakizumab administration at 100 and 200 mg was significantly more efficacious than both placebo and etanercept at week 12. [14]
The administration of intravenous tildrakizumab among patients with moderate to severe plaque psoriasis was well tolerated in all doses. [15]
Guselkumab (Tremfya)
This medication was approved by the FDA in July 2017 for the treatment of moderate to severe plaque psoriasis in adults who are potential candidates for other treatments such as phototherapy or systemic therapy. It reduces inflammation by targeting IL-23.
In patients who have been screened for susceptibility to infection and were candidates for systemic treatment or phototherapy, guselkumab significantly reduced the symptoms with a higher level of safety and efficacy. [16]
In patients with moderate to severe plaque psoriasis, guselkumab treatment improved health-related quality of life and patient-reported outcomes. [17]
Guselkumab treatment was associated with at least a 90% improvement from baseline in the Psoriasis Area and Severity Index. [18]
Brodalumab (Siliq)
This medication was FDA-approved in February 2017 for the treatment of moderate to severe plaque psoriasis. Brodalumab is recommended for patients who are candidates for phototherapy or systemic therapy and for those who do not respond to other systemic therapies. It reduces inflammation associated with psoriasis by binding to the IL-17 receptor.
In patients with moderate-to-severe psoriasis, brodalumab treatment rapidly resulted in high levels of complete and sustained skin clearance. [19]
Brodalumab treatment achieved 75% improvement in Psoriasis Area and Severity Index, suggesting that it is efficacious and safe for continuous long-term treatment of psoriasis. [20]
In patients with moderate-to-severe plaque psoriasis, brodalumab showed an acceptable safety profile and robust efficacy. [21]
Ixekizumab (Taltz)
This medication was FDA-approved in March 2016 for the treatment of moderate to severe psoriasis. It is recommended for patients who are candidates for phototherapy or systemic therapy. Ixekizumab works by targeting a protein called IL-17A.
In patients with moderate to severe plaque psoriasis, ixekizumab was statistically significantly more effective than ustekinumab at alleviating symptoms. [22]
An analysis of multiple studies found that ixekizumab produced rapid clinical improvement and a favorable short-term safety profile. [23]
Patients who received ixekizumab reported improvements in health-related quality of life, itching, and work productivity after 1 week of treatment. [24]
Adalimumab (Humira)
This medication was FDA-approved in October 2005 for the treatment of active psoriatic arthritis. In January 2008, adalimumab was approved by the FDA for the treatment of moderate-to-severe psoriasis. It suppresses inflammation by binding to TNF molecules.
In patients with chronic plaque psoriasis, adalimumab was found to be efficacious and well-tolerated. [25]
In patients with severe psoriasis and psoriatic arthritis, subcutaneous adalimumab administration at a dosage of 40 mg once a week proved to be an effective and safe treatment. [26]
The administration of adalimumab in patients with psoriasis and psoriatic arthropathy was found to be more effective than infliximab and etanercept. [27]
Etanercept (Enbrel)
In 2002, this medication was FDA-approved for the treatment of the signs and symptoms of psoriatic arthritis. In 2004, it received approval for the treatment of moderate-to-severe plaque psoriasis in adults. It works by decreasing inflammation via blockage of TNF-alpha.
Monotherapy of psoriasis with etanercept for 24 weeks produced a significant reduction in the severity of the disease and quality-of-life measures. [28]
In patients with severe recalcitrant psoriasis and psoriatic arthritis who were partially resistant to other ongoing systemic agents, the addition of etanercept produced marked improvements without toxic effects. [29]
Patients with psoriasis who received 2 weeks of etanercept therapy experienced improvement of psoriatic arthritis symptoms and reported that the treatment was generally well tolerated. [30]
Infliximab (Remicade)
In May 2005, infliximab was approved by the FDA for the treatment of psoriatic arthritis. In September 2006, the medication received approval for the treatment of chronic severe psoriasis. It works by decreasing inflammation via blockage of the inflammatory substance TNF-alpha.
In patients with moderate to severe psoriasis, infliximab treatment significantly reduced disease severity at week 6. [31]
In patients with refractory psoriasis, infliximab monotherapy produced a marked improvement in skin lesions and subjective symptoms. [32]
In patients with moderate-to-severe plaque psoriasis, the infusion of infliximab at 5 mg/kg achieved sustained improvement in symptoms compared with placebo. [33]
Calcipotriene-betamethasone dipropionate foam, 0.005%/0.064% (Enstilar)
This is a combination medication of the synthetic vitamin D3 analog calcipotriol and the synthetic corticosteroid betamethasone dipropionate. In July 2019, the FDA approved this medication for people with plaque psoriasis and adolescents between 12 and 17 years old. It works by slowing the down the growth rate of skin cells to decrease inflammation.
In patients with psoriasis vulgaris, the administration of calcipotriene-betamethasone dipropionate was associated with a significant improvement in symptoms and increased adherence. [34]
In patients with psoriasis who received treatment with calcipotriene/betamethasone dipropionate followed by 8 weeks of maintenance treatment with calcipotriene cream, a significant improvement in symptoms with a lower rate of adverse events was found. [35]
In patients with mild-to-moderate psoriasis of the body and scalp, calcipotriene/betamethasone dipropionate treatment led to a significant improvement in quality of life and a lower risk of adverse events. [36]
Results of the key randomised clinical studies investigating the efficacy of calcipotriene/betamethasone dipropionate in patients with psoriasis found that the treatment can help improve quality of life and has a high level of safety. [37]
A combination product of calcipotriene 50 microg/g and betamethasone dipropionate 0.5 mg/g showed superior efficacy with a more rapid onset of action, compared to control, in the treatment of psoriasis vulgaris. [38]
In patients with body and scalp psoriasis, the use of calcipotriene/betamethasone dipropionate topical suspension was associated with lower psoriasis-related healthcare costs, fewer psoriasis-related outpatient visits, and lesser use of systemic agents. [39]
In patients with scalp psoriasis, the use of a scalp formulation containing calcipotriol (50 μg/g) and betamethasone (0.5mg/g; as dipropionate) resulted in significant efficacy after 1 week, with a faster onset of effect than either of the individual components in the same vehicle. [40]
In patients with plaque psoriasis, treatment with a combination of topical corticosteroid and calcipotriene product resulted in greater preservation of the skin layers than topical corticosteroid use alone. [41]
Once-daily application of a topical calcipotriene + betamethasone formulation in patients with psoriasis vulgaris was associated with higher efficacy and increased patient adherence to long-term treatment compared with twice-daily use. [42]
Four weeks of treatment with calcipotriene/betamethasone dipropionate followed by 8 weeks of maintenance treatment with calcipotriene cream was found to be safe and effective in the treatment of psoriasis vulgaris. [43]
In patients with psoriasis, treatment with a fix calcipotriol/betamethasone combination was found to be more cost-effective than a non-fix morning/evening combination. [44]
Calcipotriene foam, 0.005% (Sorilux)
Calcipotriene foam 0.005% is a vitamin D analog and was the first vitamin D3 analog to be used in the treatment of psoriasis. In May 2019, the FDA approved the use of this medication for the treatment of plaque psoriasis of the scalp and body in children ages 12 to 17 years old. The following November, it was approved by the FDA for the treatment of plaque psoriasis of the scalp and body in children younger than 4 years. Calcipotriene foam 0.005% works by slowing the growth of abnormal skin cells.
In patients with psoriasis, treatment with calcipotriene ointment 0.005% was associated with a significant reduction in the disease characteristics of plaque elevation, erythema (redness), and scaling. [45]
In patients with plaque psoriasis aged 12 years and older, calcipotriene 0.005% foam administration was found to be safe and effective. [46]
In adult patients with psoriasis, calcipotriol was generally well tolerated in the short and long term with most of the adverse side effects being mild to moderate in intensity and transient. [47]
Calcipotriene 0.005% foam administration for 8 weeks was found to be safe and effective for the treatment of mild to moderate plaque-type psoriasis. [48]
Calcipotriene foam 0.005% was found to be more effective than vehicle foam in the treatment of the symptoms of scalp psoriasis over an 8-week period, with improvements observed at week 2, and had a similar safety profile to vehicle foam. [49]
Risankizumab-rzaa (Skyrizi)
This medication was approved by the FDA in April 2019 for the treatment of moderate to severe plaque psoriasis. This is indicated for affected individuals who are candidates for phototherapy or systemic therapy. It decreases inflammation by blocking the action of interleukin-23 (IL-23).
Patients with moderate to severe plaque psoriasis who received treatment with risankizumab had improved health-related quality of life and reported higher tolerability, both in the short- and longer-term. [50]
In patients with active psoriatic arthritis who have responded inadequately or are intolerant to conventional synthetic disease-modifying antirheumatic drugs, risankizumab treatment produced significantly greater improvement of signs and symptoms and was associated with higher tolerability compared with placebo. [51]
Four pivotal Phase III trials found that risankizumab was safe and effective in patients with moderate-to-severe plaque psoriasis. [52]
Ustekinumab (Stelara)
This medication was approved by the FDA on October 2017 for the treatment of moderate to severe plaque psoriasis in patients who are candidates for phototherapy or systemic therapy. It is also indicated for adolescents 12 years old and up who have plaque psoriasis. Ustekinumab works by decreasing inflammation associated with psoriasis via blockage of the inflammatory proteins IL-12 and IL-23.
In patients with moderate to severe plaque psoriasis, the administration of subcutaneous ustekinumab 45 or 90 mg (administered as two injections 4 weeks apart) achieved a 75% improvement on the Psoriasis Area and Severity Index (PASI 75) score at 12 weeks compared to placebo treatment. [53]
Ustekinumab has demonstrated efficacy, short-term safety, and convenience of use in the treatment of plaque psoriasis and psoriatic arthritis. [54]
In pediatric patients (≥ 6 to < 12 years of age) with moderate-to-severe psoriasis, ustekinumab produced a positive change in the Children’s Dermatology Life Quality Index (CDLQI) and significantly reduced mean serum concentrations of interleukin-17A/F and interleukin-22. [55]
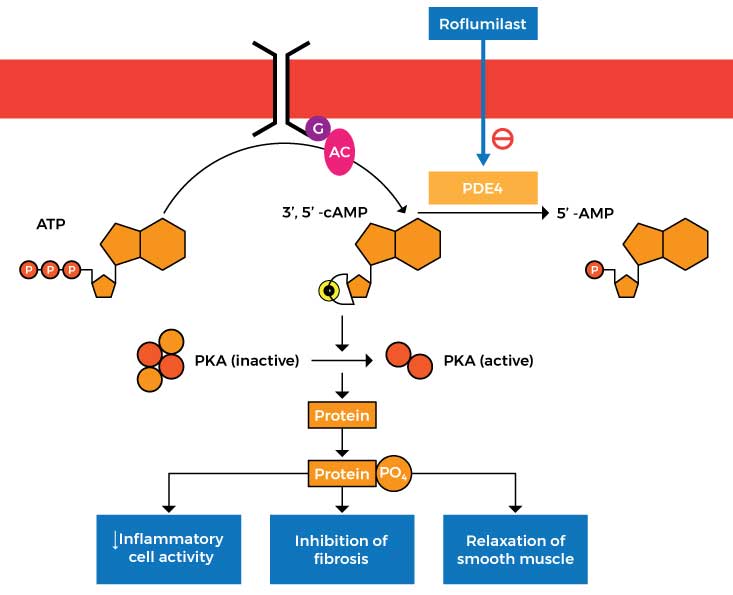
Zoryve (Roflumilast) Cream 0.3%
This medication was approved by the FDA on July 29, 2022 for the treatment of plaque psoriasis in patients aged 12 years and older. It works by inhibiting phosphodiesterase-4 (PDE4), an enzyme responsible for the production of inflammatory substances.
In patients with chronic plaque psoriasis, roflumilast cream administration was safe and highly effective at improving signs and symptoms at doses of 0.5% and 0.15%. [56]
Roflumilast cream administered once daily to affected areas of chronic plaque psoriasis demonstrated superior efficacy to vehicle cream in achieving a state of clear or almost clear at 6 weeks. [57]
In patients with psoriasis vulgaris, topical treatment with a cream formulation of roflumilast resulted in decreased inflammation (as evidenced by a decrease in skin infiltrate thickness) and reduced psoriasis severity. [58]
Non-FDA-Approved Treatment for Psoriasis
KPV (Lysine-Proline-Valine)
KPV is a tripeptide (Lysine-Proline-Valine) that possesses potent anti-inflammatory properties. It’s a C-terminal tripeptide of α-MSH (alpha-Melanocyte-stimulating hormone). Peptides like KPV often act as hormones and relay information from one tissue through the blood to another via biological messengers.
Interestingly, KPV appears to only have an effect in the setting of inflammation and it has almost no effect in normal tissue. The main reason for this is that KPV enters the cells using a transporter that is unregulated in case of inflammation. This suggests that the peptide may serve as an effective or preventive medication in inflammatory conditions like psoriasis. If taken regularly, KPV will be available when necessary and the body can simply excrete it if inflammation and other symptoms are not present.
Once psoriasis begins to resolve, it can leave behind dark spots known as hyperpigmentation. The reason behind this is that psoriasis causes the skin to produce higher levels of inflammatory chemicals. This in turn affects the production of the skin pigment known as melanin, resulting in hyperpigmentation. Another amazing advantage of KPV is that, unlike alpha-Melanocyte-stimulating hormone, it does not cause skin pigmentation. [59] This means that KPV can help treat psoriasis by decreasing inflammation without aggravating hyperpigmentation associated with the disease.
In an imiquimod-induced psoriasis mouse model, the application of a gel containing alpha-MSH resulted in a reduction in psoriasis-associated inflammation. [60] The treatment also significantly decreased psoriatic-like plaque skin sections.
KdPT
KdPT is a tripeptide derivative of α-melanocyte–stimulating hormone (α-MSH or alpha-MSH) which has a similar structure to KPV. Like KPV, KdPT has potent anti-inflammatory effects that can help treat the signs and symptoms of psoriasis.
In murine and human skin affected by psoriasis, KdPT treatment significantly reduced hyperkeratosis (thickening of the outer layer of the skin) and acanthosis (dark skin discoloration in body folds and creases). [61]
In mice with imiquimod-induced psoriasis-like skin lesions, KdPT treatment alleviated the lesions by inhibiting the inflammatory response. [62]
Alpha-MSH was shown to inhibit disease progression in a mouse model of psoriasis-like skin inflammation and prevented the activation and multiplication of effector T cells (attack healthy cells) in subjects with psoriasis. [63]
Bimekizumab
This injectable biologic drug is being tested as a potential treatment for chronic plaque psoriasis. It decreases inflammation by blocking IL-17. Recent studies have found that bimekizumab is safe and effective in patients with psoriasis.
In patients with moderate-to-severe psoriasis, bimekizumab treatment resulted in greater skin clearance over 16 and 48 weeks compared with secukinumab. [64]
Phase 3 trials showed that bimekizumab produced a safety profile similar to the other biologic drugs tested and had higher tolerability. [65]
BE READY, a phase 3 clinical trial, showed that bimekizumab showed high levels of response and was well tolerated by patients with moderate to severe plaque psoriasis. [66]
Janus Kinase Inhibitors (JAK) Inhibitors
Also known as DMARDs (disease-modifying antirheumatic drugs), they are a group of disease-modifying drugs indicated for the treatment of psoriatic arthritis. They work by specifically targeting pathways involved in the production of inflammatory proteins.
In both phase 2 and 3 trials, JAK inhibitors have shown clinical efficacy as measured by the Psoriasis Area and Severity Index 75 response and were associated with higher tolerability. [67]
JAK inhibitors proved to be effective in improving the symptoms of psoriatic arthritis and enhancing the quality of life of patients. [68]
JAK inhibitors were found to have a lower prevalence of side effects compared to other biologic drugs for psoriasis. [69]
6-Thioguanine
This medication is indicated for the treatment of moderate to severe plaque-type psoriasis. It works by stopping the growth of malignant cells by disrupting DNA and RNA.
In patients who have failed to respond to other systemic agents, 6-Thioguanine was found to be an effective treatment. [70]
In patients with moderate to severe plaque-type psoriasis, 6-Thioguanine was found to be safe and effective when administered for defined periods and with careful hematologic monitoring. [71]
In patients with severe recalcitrant psoriasis, 78% of the subjects who received 6-Thioguanine treatment had a dramatic improvement in symptoms. [72]
Azathioprine
This medication is indicated for the treatment of chronic plaque psoriasis. It works by suppressing activated T lymphocyte cells, which in turn stops the production of inflammatory substances.
In patients with chronic plaque psoriasis, azathioprine 300 mg weekly pulse was effective in achieving Psoriasis Area and Severity Index (PASI) of 75 in 42% of the subjects. [73]
Weekly azathioprine pulse appears to be beneficial in treating chronic plaque psoriasis, however, it is less effective than weekly methotrexate. [74]
In patients with psoriasis, azathioprine pulse therapy was associated with a decrease in or disappearance of signs and symptoms of the disease for more than five years. [75]
Fumaric Acid Esters
These are small molecules with immunomodulating, anti-inflammatory, and anti-oxidative properties. They work by reducing the levels of inflammatory substances in the body, making them an effective treatment for moderate to severe psoriasis.
In patients intolerant of or unresponsive to other agents, the use of fumaric acid esters was found to be safe and effective in the management of moderate to severe psoriasis. [76]
In patients with moderate to severe psoriasis, fumaric acid ester treatment was associated with a marked improvement in symptoms after 6 months. [77]
Fumaric acid esters in combination with phototherapy induced a faster therapeutic response with higher tolerability in patients with moderate-to-severe plaque psoriasis. [78]
Mirikizumab
This is a humanized IgG4 monoclonal antibody indicated for the treatment of psoriasis. It works by binding to the inflammatory substance interleukin 23.
Patients treated with mirikizumab 300 mg at 8-week intervals achieved a 90% improvement in the Psoriasis Area and Severity Index (PASI 90) response at week 16. [79]
The administration of mirikizumab in patients with chronic plaque psoriasis was associated with the suppression of inflammatory markers. [80]
Mirikizumab was superior to placebo at week 16 and was associated with higher efficacy and tolerability. [81]
Fluocinonide
This topical steroid helps relieves itching, redness, dryness, crusting, scaling, inflammation, and other discomforts in patients with psoriasis. It works by preventing the release of inflammatory substances in the body.
In patients with refractory psoriasis that had responded poorly to previous therapy, fluocinonide gel (0.05 percent) treatment for 6 weeks completely cleared the lesions. [82]
Fluocinonide treatment was associated with 60-80% clearing of lesions compared to zero with the placebo. [83]
Hydroxyurea
This is an oral cancer medication that is found to be effective in the treatment of psoriasis. It works by reducing the levels of inflammatory molecules.
In patients with psoriasis, the administration of low-dose hydroxyurea was associated with a significant improvement in the Psoriasis Area and Severity Index (PASI) scores. [84]
A study involving patients with chronic plaque psoriasis reported that hydroxyurea is an effective and reasonably safe second-line agent for psoriasis. [85]
In patients with extensive chronic plaque psoriasis, hydroxyurea treatment produced an adequate response as evidenced by a 35% reduction in the Psoriasis Area and Severity Index at or before 8 weeks. [86]
Mycophenolate mofetil
This is an immunosuppressant medication that works by inhibiting activated lymphocytes. As a result, the immune system does not attack healthy skin cells by mistake.
In a man with severe psoriasis, Mycophenolate mofetil improved the psoriasis area and severity index score without short-term side effects. [87]
Mycophenolate mofetil was found to be a good alternative for the treatment of psoriasis in patients who are unable to take methotrexate. [88]
In patients with severe psoriasis, Mycophenolate mofetil was found to be safe and effective. [89]
Sulfasalazine
This is a disease-modifying antirheumatic drug (DMARD) that works by reducing the levels of inflammatory substances.
Sulfasalazine treatment resulted in a significant improvement in inflammatory indices (C-reactive protein and erythrocyte sedimentation rate levels) in patients with active psoriatic arthritis. [90]
The combination of sulfasalazine and pentoxifylline produced a good response in cases of extensive psoriasis. [91]
Sulfasalazine appears to be an effective second-line agent for the treatment of psoriatic arthritis and was found to suppress the immune function by altering B cell number and function. [92]
Tacrolimus
This medication is indicated for eczema and psoriasis. Tacrolimus works by suppressing the immune system to prevent damage to the skin cells.
In patients with facial or intertriginous psoriasis, Tacrolimus treatment was associated with a reduction in disease severity. [93]
In patients with psoriasis on the face and intertriginous areas, Tacrolimus resulted in a significant improvement in the physician’s assessment of the individual signs and symptoms. [94]
Oral tacrolimus has been shown to exert a higher efficacy in the treatment of severe, refractory psoriasis. [95]
Topical Calcineurin Inhibitors
These medications work by altering the immune system, thus, decreasing inflammation associated with psoriasis.
A review of multiple studies demonstrated the efficacy of topical calcineurin inhibitors in the treatment of facial, genital, and intertrigious psoriasis. [96]
A study also found that topical calcineurin inhibitors can significantly reduce facial, genital, and intertriginous psoriatic lesions. [97]