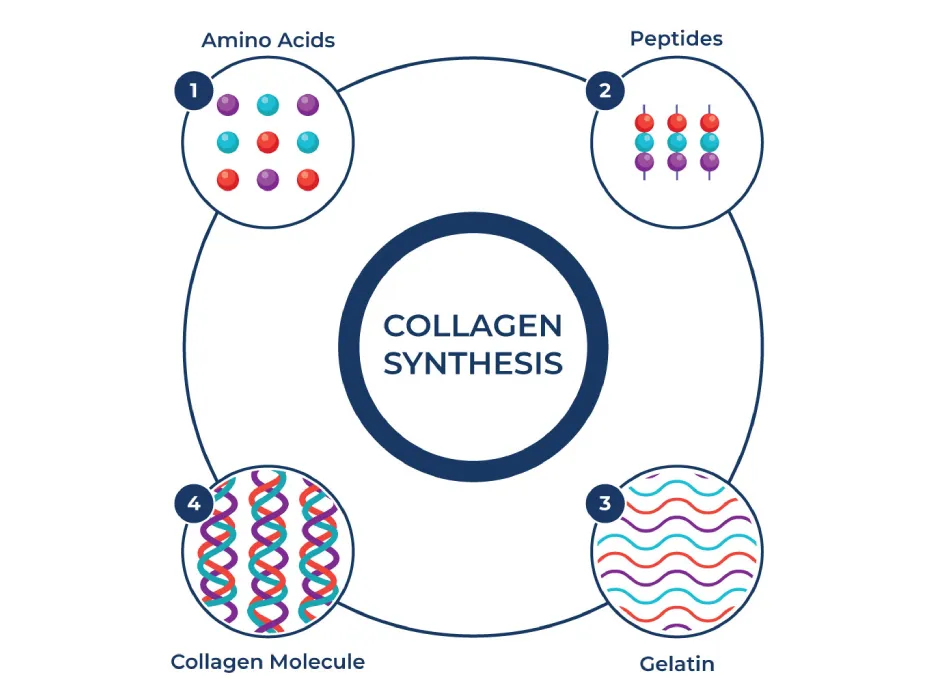
Potential Health Benefits
Vitamin D benefits include supporting bone health by enhancing calcium absorption, boosting immune function, and improving muscle strength. It also plays a role in regulating mood and reducing inflammation.
- Improves bone health [1-86]
- Strengthens the immune system [88-162]
- Fights cancer [163-270]
- Maintains a healthy heart [271-338]
- Boosts cognitive function [353-393]
- Improves muscle mass and strength [394-425]
- Wards off depression [426-489]
- Improves blood sugar levels [490-533]
- Improves blood pressure [534-600]
- Improves cholesterol levels [601-628]
- Helps lose weight [629-683]
- Improves sleep quality [699-708]
- Keeps kidneys healthy [709-770]
- Improves sexual function [771-789]
- Improves fertility [790-840]
- Prevents hair loss [841-873]
Key Takeaways
- Supports Bone Health: Vitamin D helps the body absorb calcium, which is crucial for maintaining strong bones and preventing conditions like osteoporosis.
- Boosts Immune Function: It plays a vital role in strengthening the immune system, helping to defend against infections and chronic diseases.
- Improves Muscle Function: Adequate vitamin D levels are important for maintaining muscle strength and reducing the risk of falls, especially in older adults.
- Regulates Mood: Vitamin D has been linked to improved mood and may help alleviate symptoms of depression.
- Sunlight and Diet Sources: The body can produce vitamin D through sun exposure, but it can also be obtained from food sources like fatty fish and fortified products, or through supplements.
What is Vitamin D?
Vitamin D, also known as cholecalciferol (vitamin D3), ergocalciferol (Vitamin D2), or the sunshine vitamin, is one of the 24 micronutrients critical for human survival. In spite of the name, vitamin D is often referred to as a pro-hormone because the body is capable of producing its own vitamin D through mild sun exposure, while vitamins are nutrients that cannot be produced by the body and must be acquired by eating vitamin-rich foods or taking supplements.Vitamin D: Essential Benefits, Deficiency Risks, and Best SourcesVitamin D: Essential Benefits, Deficiency Risks, and Best Sources
Vitamin D Deficiency
It is estimated that sensible sun exposure on bare skin for 5-10 minutes twice or thrice a week allows the body to produce adequate amounts of vitamin D. However, this vitamin has a half-life of only 2 weeks, meaning that vitamin D stores can be depleted. Recent studies have suggested that vitamin D deficiency affects almost 50% of adults and children worldwide. [252] Because of the extensive benefits of vitamin D for human health, the United States Department of Agriculture (USDA) recently increased the recommended daily allowance for vitamin D for adults and the American Academy of Pediatrics recommends that children get adequate vitamin D.
The following are the recommended intakes of vitamin D throughout life: [253]
- Infants 0-12 months – 400 IU (10 mcg)
- Children 1-18 years – 600 IU (15 mcg)
- Adults to age 70 – 600 IU (15 mcg)
- Adults over 70 – 800 IU (20 mcg)
- Pregnant or lactating women – 600 IU (15 mcg)
Causes of Vitamin D Deficiency
There are certain medical conditions and factors that can cause a deficiency in vitamin D. These include the following:
- Kidney and liver diseases: These medical conditions decrease the production of an enzyme needed to convert vitamin D to its usable form.
- Cystic fibrosis, Crohn’s disease, and celiac disease: These diseases prevent the intestines from absorbing adequate amounts of vitamin D.
- Gastric bypass surgery: This medical procedure involves the removal of a part of the stomach and/or the intestines in order to reduce food intake and achieve significant weight loss. However, this weight-loss surgery impairs vitamin D absorption.
- Obesity: High amounts of fat hold onto the vitamin D, preventing it from being released into the bloodstream.
- Age: With advancing age, the skin’s ability to produce adequate amounts of vitamin D lessens.
- Skin color: People with dark-colored skin produce lesser amounts of vitamin D than those with fair-colored skin.
- Certain medications: Laxatives, steroids, seizure medications, weight-loss drugs, and cholesterol-lowering drugs decrease vitamin D levels in the body as a side effect.
Vitamin D3 Deficiency Symptoms
In men and women with vitamin D deficiency, the symptoms are generally subtle so they may not notice them easily, even if it’s having a significant impact in their quality of life. The common signs and symptoms of vitamin D deficiency are the following:
- Anxiety
- Bone loss
- Chronic pain
- Depression
- Erectile dysfunction
- Extreme fatigue
- Getting sick or infected often
- Hair loss
- High blood pressure
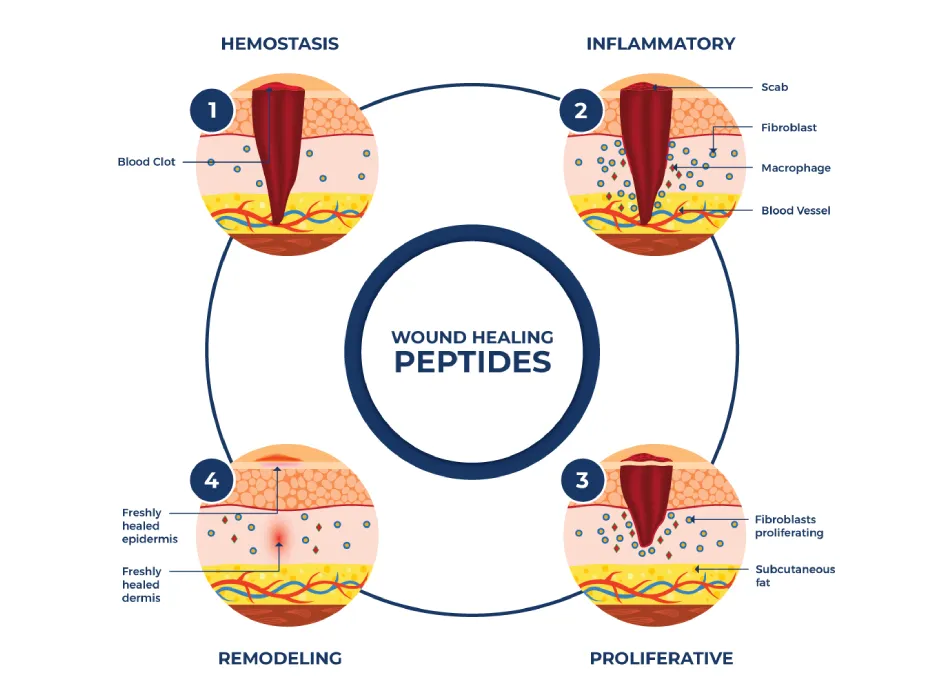
- Impaired wound healing
- Inflammation and swelling
- Joint and muscle pain
- Reduced libido
- Sleeping difficulties
If you are suffering from symptoms of vitamin D deficiency, it is recommended to consult immediately with your doctor to get your vitamin D levels checked. A detailed physical examination will also help rule out any medical conditions, risk factors for vitamin D deficiency, and other factors that may affect vitamin D levels. If left untreated, severely low levels of vitamin D can lead to osteomalacia in adults and rickets in children. Both of these diseases can lead to soft and weak bones, bone and muscle pain, and a higher incidence of fractures.
Vitamin D Food Sources
Sunlight is the most cost-effective way of boosting vitamin D stores. However, for people living in northern latitudes and for dark-skinned people, getting adequate amounts of vitamin D through sun exposure can be a problem. Fortunately, they can still load up on this vitamin by consuming the following foods:
- Beef liver
- Cheese
- Cod liver oil
- Egg
- Fortified skim milk
- Herring
- Raw maitake mushrooms
- Salmon
- Sardines
- Swordfish
- Tuna
- Yogurt
Research on Vitamin D
A. Improves Bone Health

Vitamin D plays a major role in improving bone strength and quality. Without the “sunshine vitamin”, our bodies cannot effectively absorb calcium and phosphorus – two key nutrients in bone growth and development. There is increasing evidence that vitamin D supplementation may help improve bone health in the older population as well as those with medical conditions affecting the bones:
- Studies show that people with low blood levels of vitamin D tend to suffer from more bone loss as evidenced by a decrease in bone mineral density (BMD). [1-32]
- In women with postmenopausal osteoporosis, vitamin D replacement therapy is necessary to maximize the response to medications that prevent bone breakdown. [33-39]
- Research shows that taking vitamin D supplements may lower the risk of bone fractures by 22-33%. [40-41]
- In elderly or postmenopausal women, vitamin D supplementation significantly prevents fractures and improves fracture healing. [42-43]
- In the elderly, vitamin D supplementation significantly reduces the risk of falls. [44-47]
- In patients with Parkinson’s disease, two years of 1,000 IU of vitamin D2 may help prevent hip fractures and preserve BMD. [48]
- When combined with calcium, vitamin D supplementation significantly lowers the risk of fractures. [49-52]
- In patients with osteoporosis, vitamin D replacement therapy significantly increases BMD at different skeletal sites. [53-86]
B. Strengthens the Immune System
Vitamin D plays a major role in the regulation of immune function by turning on or off genes and processes necessary for optimum health. In fact, vitamin D is touted as a “miracle nutrient” for the immune system as it enables the body to produce hundreds of antimicrobial peptides, which help ward off various infections and diseases. An overwhelming body of clinical evidence supports the immune-modulating properties of vitamin D:
- In patients with asthma, daily vitamin D doses of 7.5–30 mcg (300–1,200 IU) decreases asthma attacks. [87-88]
- Lower levels of vitamin D are strongly linked with an increased risk of respiratory tract infections. [89-91]
- Vitamin D supplementation significantly reduces the risk of upper respiratory tract infections (URTI). [92-93]
- Vitamin D strengthens the immune system by boosting mucosal defenses and preventing excessive inflammation. [94]
- Vitamin D has strong antimicrobial activity that is effective against a broad range of bacteria and viruses, including M. tuberculosis and influenza virus. [95-99]
- In older adults with infections, high-dose vitamin D supplementation significantly reduces antibiotic use by as much as 50%. [100-101]
- In patients with chronic obstructive pulmonary disease (COPD), high doses of vitamin D improve lung function, rates of exacerbation, morbidity, and mortality. [102]
- In adults with cystic fibrosis and worsening pulmonary symptoms, a single 250,000 IU dose of vitamin D significantly increases hospital-free days. [103-104]
- Vitamin D supplementation is associated with a 13% decreased risk of repeat pneumonia. [105]
- In patients with tuberculosis, vitamin D supplementation decreases markers of the disease and improves clinical outcomes. [106-118]
- In patients with multiple sclerosis and rheumatoid arthritis, higher vitamin D levels appear to slow disease activity. [118-121]
- Vitamin D affects T cell maturation and facilitates the induction of T regulatory cells. [122-126]
- Vitamin D also has effects on monocytes and dendritic cells (DCs) of the immune system. [127-131]
- In patients with active lupus, vitamin D significantly decreases both spontaneous and stimulated immunoglobulin production from B cells, interferon (IFN), and other immune cells, thus preventing excessive immune response. [132-137]
- In patients with multiple sclerosis, vitamin D inhibits the excessive production of CD4 cells. [138]
- Vitamin D can also suppress an excessive immune response, thus restoring normal immune function. [139-162]
C. Fights Cancer

Vitamin D is extremely important for regulating the growth of billions of cells, enhancing their communication, and modulating general cellular function. Aside from these roles, vitamin D also has potent anti-cancer properties that can help eradicate malignant cells in the body. There is very strong clinical evidence that vitamin D supplementation may help protect against various types of cancers in both men and women:
- The majority of studies found a protective relationship between higher levels of vitamin D and a lower risk of colorectal cancer. [163-184]
- On the other hand, lower vitamin D levels are associated with an increased risk of cancers of the colon, breast, and prostate as well as other cancers. [185-188]
- Adequate vitamin D levels can reduce bladder cancer risk and related death by 25%. [189-191]
- Higher vitamin D levels also correlate with lower lung cancer risk and better prognosis. [192-197]
- Higher vitamin D levels are also associated with a lower risk of skin cancer. [198-199]
- Sunlight exposure has been found to significantly reduce the likelihood of various types of cancers (bladder, brain, breast, colon, cervical, liver, lung, ovarian, pancreas, pleura, prostate, rectal, and thyroid cancer as well as non-Hodgkin’s lymphoma). [220-204]
- In human cancer cell lines, vitamin D administration prevents cancer cell progression by inducing programmed cell death (apoptosis). [205-214]
- Vitamin D has a chemopreventive mechanism that can help eradicate cancer cells. [215]
- In patients with lung cancer, vitamin D inhibits signaling pathways that promote the spread of cancer cells (metastasis) and cell division. [216-219]
- Vitamin D inhibits prostaglandin synthesis, which in turn prevents the growth and reproduction of tumor cells. [220]
- Vitamin D inhibits tumor angiogenesis (formation of new blood vessels), thereby preventing its progression. [221-226]
- In palliative cancer patients, vitamin D supplementation is safe and effective in improving pain management (1 month after treatment) and decreasing infections (3 months after treatment). [227]
- In postmenopausal women with breast cancer, vitamin D supplementation counters the detrimental effects of chemotherapeutics on bone health. [228-230]
- In patients with leukemia and lymphoma, vitamin D replacement therapy improves survival. [231-235]
- In men with low-risk prostate cancer, daily supplementation with 4000 IU of vitamin D for 1 year decreases the number of biopsy-positive cores (predictor of clinical outcome in prostate cancer). [236]
- In men with prostate cancer, oral vitamin D supplementation at a dose of 40,000 IU reduces cancer cell proliferation marker Ki67. [237]
- In patients with advanced breast and colorectal cancer, administration of the vitamin D analogue EB 1089 stabilizes the disease. [238]
- In patients with advanced solid tumors, oral administration of the vitamin D3 analogue ILX23-7553 is associated with tumor growth inhibition. [239]
- In breast cancer patients with bone metastases, high-dose (10,000 IU/day) vitamin D supplementation significantly decreases the number of pain sites over time. [240]
- In women with breast and ovarian cancer, vitamin D supplementation could help maintain bone health and enhance cancer survival rate. [241]
- Vitamin D deficiency has been associated with an increased risk of cancer, and supplementation may play a supportive role in cancer prevention and treatment. [242-269]
- Preliminary results from an ongoing clinical trial, the VITamin D and OmegA-3 TriaL, show that vitamin D supplementation may help improve cancer symptoms and survival rate. [270]
D. Maintains a Healthy Heart
Vitamin D can also help reverse heart damage and protect against cardiovascular diseases. Because it has beneficial effects on various cardiovascular parameters such as blood pressure, blood sugar, and cholesterol levels, vitamin D can be an important nutrient for the maintenance of a healthy heart. A growing number of studies support the cardiovascular effects of vitamin D:
- Vitamin D deficiency significantly increases one’s risk of developing cardiovascular disease and related deaths. [271-290]
- In adults with cardiovascular disease, a very modest amount of vitamin D (500 IU) is associated with an 8% reduction in all-cause mortality. [291-292]
- Vitamin D helps improve the recovery of the cells of the heart (cardiomyocytes) after an injury, suggesting that it can help reverse damage. [293]
- In patients with heart failure, vitamin D supplementation significantly reduces inflammatory cytokines (involved in the development of heart disease). [294-297]
- In patients with chronic heart failure, one year of 4,000 IU/day of vitamin D3 is associated with a 6.1% improvement in ejection fraction (percentage of blood leaving the heart during each contraction). [298]
- Vitamin D prevents cardiovascular disease by inhibiting plaque formation in the heart. [299]
- Vitamin D is involved in the regulation of growth and reproduction of smooth muscle cells and cardiomyocytes. [300-302]
- In frail elderly patients, vitamin D supplementation is associated with a 7% significant mortality reduction. [303]
- In hemodialysis patients and those with heart failure, vitamin D supplementation reduces natriuretic peptide (a diagnostic marker of heart failure) and left ventricular mass, as well as improves left ventricular function. [304-310]
- In patients with heart failure, vitamin D supplementation decreases disease severity as evidenced by a significant reduction in natriuretic peptide levels and an increase in walking distance. [311]
- In hemodialysis patients, vitamin D supplementation reduces important surrogate markers of cardiovascular risk such as systemic inflammation and left ventricular hypertrophy. [312]
- Early intervention with vitamin D supplementation is an ideal approach to preventing childhood cardiovascular disease. [313]
- Vitamin D3 prevents heart disease by regulating the inflammatory response in human coronary arterial endothelial cells. [314]
- In patients with heart disease and elderly people, vitamin D administration is associated with a statistically significant reduction in mortality and cardiovascular risk. [315-319]
- In the elderly population, restoration of vitamin D levels to normal significantly improves heart muscle contractility and walking capacity. [320]
- In patients suffering from heart failure, vitamin D supplementation improves functional activity and severity of congestive heart failure. [321-322]
- Vitamin D deficiency has been associated with an increased risk of cardiovascular disease, and supplementation may play a role in its prevention and management. [323-338]
E. Boosts Cognitive Function

Cognitive dysfunction can lead to confusion, memory problems, and difficulty concentrating. Interestingly, scientists found that vitamin D can function as a neurosteroid or “cognitive enhancer”, thus preventing age-related cognitive decline and improving overall mental health. A growing body of scientific evidence supports the brain-boosting properties of vitamin D:
- Vitamin D deficiency is associated with worse cognitive performance and impairment.[339-352]
- In older adults, 16 months of 800 IU/day vitamin D3 supplementation improves cognitive performance. [353]
- Vitamin D supplementation decreases the risk of Alzheimer’s disease. [354]
- In younger individuals, vitamin D supplementation improves executive functioning and mental health. [355]
- In individuals with inadequate baseline vitamin D levels, higher doses of vitamin D supplementation improve nonverbal (visual) memory. [356]
- In frail elder adults, vitamin D supplementation improves cognition. [357]
- In Alzheimer’s patients over the age of 60 years, vitamin D supplementation decreases plaque build-up in the brain. [358]
- In patients with severe vitamin D deficiency, vitamin D supplementation improves learning and memory. [359]
- In older adults, vitamin D deficiency is associated with dementia, and vitamin D supplementation might have a protective effect. [360-393]
F. Improves Muscle Mass and Strength

Muscle mass and strength tend to decrease with advancing age, primarily as a result of reduced physical activity. This loss of muscle mass, medically known as sarcopenia, has a negative impact on one’s ability to perform activities of daily living. Fortunately, vitamin D supplementation may be an effective method of countering age-related muscle wasting and those caused by chronic medical conditions. There is an overwhelming body of clinical research supporting the muscle-enhancing properties of vitamin D:
- In patients on hemodialysis, vitamin D treatment is associated with greater muscle size and strength. [394]
- In patients with end-stage renal disease (ESRD), vitamin D treatment improves muscle function. [395-398]
- In vitamin D deficient patients, vitamin D supplementation restores muscle mass and strength. [399-401]
- In patients with amyotrophic lateral sclerosis (ALS), vitamin D supplementation improves scores in a series of tests assessing muscle function. [402]
- In women, higher blood levels of vitamin D are associated with better results in musculoskeletal function. [403]
- In patients aged 65 years and above, higher vitamin D levels are associated with increased handgrip strength. [404]
- In people aged 70 years and above, a weekly dose of 8400 IU vitamin D3 improves postural stability, muscle strength and safety. [405]
- In professional male and female elite ballet dancers, 4 months of 2,000 IU of vitamin D3 administration improves muscle strength. [406]
- In athletes, vitamin D enhances the recovery of muscles shortly after intense exercise. [407]
- In professional soccer players, vitamin D3 supplementation improves musculoskeletal performance measures such as sprint times and vertical jump height. [408-409]
- In elite ballet dancers, vitamin D supplementation significantly increases muscle strength and musculoskeletal performance. [410]
- In patients with sarcopenia, vitamin D supplementation improves muscle strength, muscle size, and neuromuscular performance. [411-412]
- Patients with higher vitamin D blood levels have better muscle performances of the lower extremities than patients with lower levels. [413-415]
- In healthy males who underwent muscle damage through eccentric quad exercises, 4000 IU vitamin D administration improves muscle recovery and strength. [416]
- In overweight and obese adults, vitamin D supplementation during resistance training induces an early improvement in peak power (a measure of muscle strength). [417]
- Studies demonstrate that vitamin D supplementation can significantly enhance muscle strength and improve mobility. [418-425]
G. Wards off Depression
A healthy brain is the first line of defense against mental health problems including depression. Since vitamin D plays a major role in brain health throughout all stages of life, boosting its levels may help increase energy levels and improve overall mood. Several lines of evidence support the antidepressant effect of vitamin D:
- Vitamin D increases the amount of serotonin, a brain chemical that contributes to feelings of well-being and happiness. [426-427]
- Low blood levels of vitamin D are associated with an increased risk of depression and low mood. [428-454]
- In healthy individuals, vitamin D3 at a dose of 400-800 IU per day is associated with more positive emotions. [455]
- In women with lower blood levels of vitamin D, supplementation improves scores in the Beck Depression Inventory-II test. [456-457]
- In patients with seasonal affective disorder (depression associated with late autumn and winter), vitamin D supplementation improves all outcome measures. [458]
- In vitamin D-deficient patients with depression, vitamin D supplementation is safe and effective in reducing depressive symptoms when given as an adjunctive treatment. [459]
- In adolescent girls, high-dose vitamin D supplementation is associated with a reduction in depression scores. [460]
- In depressed adolescents, vitamin D3 supplementation over 3 months ameliorates depressive symptoms. [461]
- In patients with clinically significant depression, vitamin D supplementation effectively reduces depressive symptoms. [462]
- In patients with major depressive disorder, vitamin D supplementation improves scores on the Hamilton Depression Rating Scale (HDRS) and Beck Depression Inventory (BDI). [463]
- In depressed patients with vitamin D deficiency, a single injection of a high dose of vitamin D significantly improves depressive symptoms. [464]
- In overweight and obese men and women, supplementation with high doses of vitamin D seems to ameliorate depressive symptoms. [465]
- In adults, vitamin D supplementation significantly improves well-being without any adverse side effects. [466]
- In women with type 2 diabetes, vitamin D supplementation improves mood and health status. [559]
- In people with depression, vitamin D replacement therapy could improve health outcomes and quality of life. [467]
- In patients with significant depressive symptoms or depressive disorder, vitamin D supplementation dramatically reduces symptoms. [468-489]
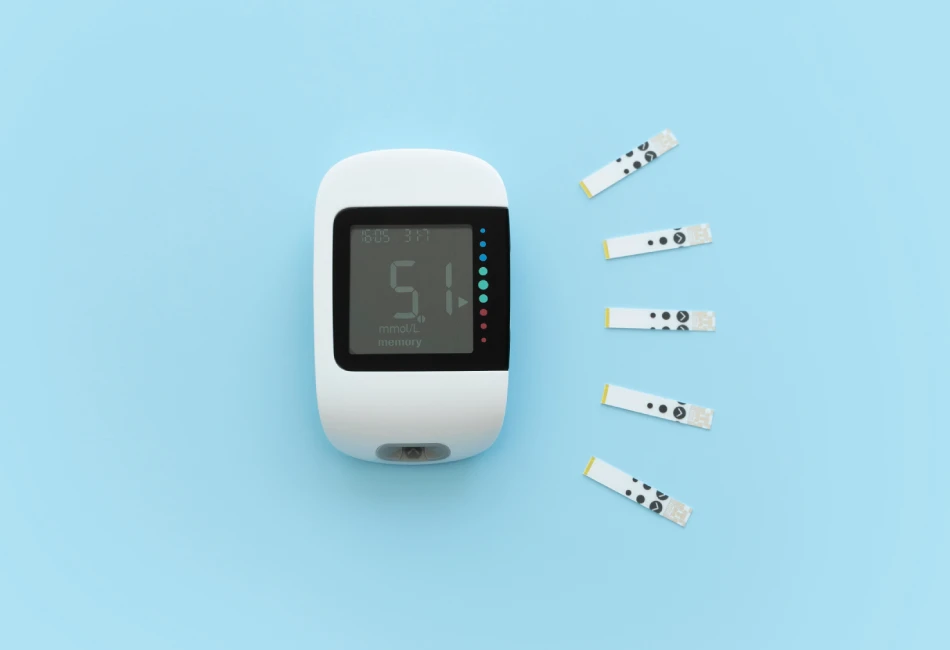
H. Improves Blood Sugar Levels
Vitamin D is believed to help lower blood sugar levels as well as diabetes risk by improving the body’s response to the effects of insulin – the hormone that regulates blood sugar levels. Of further interest, vitamin D status is associated with blood sugar levels, suggesting that restoration of vitamin D to healthy levels may have beneficial effects. An overwhelming body of clinical research supports the anti-diabetic properties of vitamin D:
- Vitamin D deficiency is associated with a higher prevalence of type 2 diabetes and higher blood sugar levels. [490-509]
- Vitamin D may help protect against type 2 diabetes by preventing insulin resistance, increasing insulin sensitivity (the body’s response to the effects of insulin), and enhancing the function of the cells responsible for producing insulin. [510-512]
- In patients with type 2 diabetes, vitamin D3 supplementation increases insulin secretion by 34.3%. [513]
- In adults at risk for type 2 diabetes, vitamin D supplementation improves insulin secretion and reduces HbA1c, a three-month average measure of blood sugar levels. [514]
- In African American diabetics, vitamin D supplementation reduces the rate of insulin sensitivity impairment by improving pancreatic insulin secretion. [515]
- In subjects with vitamin D deficiency and impaired fasting glucose, supplementation with 10,000 IU of vitamin D daily for 4 weeks is associated with an increase in insulin sensitivity. [516]
- In adults with type 2 diabetes, vitamin D treatment is associated with a modest reduction of HbA1C. [517]
- In patients with type 2 diabetes, vitamin D supplementation has beneficial effects on blood sugar balance and insulin sensitivity. [518]
- In patients with gestational diabetes, vitamin D supplementation improves blood sugar metabolism. [519]
- In vitamin D-deficient or non-obese type 2 diabetes patients, vitamin D supplementation is safe and effective at improving blood sugar control. [520]
- In pregnant women with gestational diabetes, vitamin D supplementation significantly decreases blood sugar levels. [521]
- Vitamin D appears to have a “balancing effect” on blood sugar levels. [522]
- In prediabetic individuals, vitamin D supplementation improves blood sugar measures and insulin sensitivity. [523]
- In patients with type 2 diabetes, vitamin D supplementation decreases high blood sugar levels by improving beta cell secretion and insulin sensitivity. [524]
- In patients with type 2 diabetes and in non-diabetics, vitamin D supplementation lowers fasting plasma glucose (FPG) and HbA1c. [525]
- In obese hypertensive patients, vitamin D supplementation reduces HbA1c levels. [526]
- In adults who are at high risk for diabetes type 2, vitamin D supplementation improves blood sugar control. [527]
- In patients with type 2 diabetes, daily consumption of vitamin D- or vitamin D + calcium-fortified yogurt drink improves blood sugar levels. [528]
- When combined with calcium, vitamin D may help improve blood sugar metabolism and significantly lower the risk of diabetes. [529-531]
- Vitamin D lowers blood sugar levels by facilitating blood sugar transportation into different organs and regulating insulin receptor genes and insulin secretion. [532-533]
I. Improves Blood Pressure
High blood pressure, medically known as hypertension, can significantly increase one’s risk of developing chronic, debilitating medical conditions such as heart disease, diabetes, stroke, and other fatal diseases. By reducing blood pressure, the risk of getting these diseases also decreases. There is strong scientific evidence that vitamin D has potent anti-hypertensive properties that can help ward off diseases associated with high blood pressure:
- Low levels of vitamin D are strongly linked with hypertension. [534-546]
- One study found that for every increase in vitamin D supplementation and vitamin D levels in the body, systolic blood pressure declined. [547]
- In people who have low levels of vitamin D, supplementation appears to be more effective in lowering blood pressure. [548]
- In hypertensive patients, vitamin D supplementation at a dose of 25,000 IU per week for 8 weeks reduces the risk of hypertension. [549]
- Higher levels of vitamin D are associated with a significant reduction in blood pressure. [550-554]
- In patients with type 2 diabetes, vitamin D supplementation reduces blood pressure by improving blood vessel function. [555]
- In patients with vitamin D insufficiency, supplementation significantly reduces central systolic blood pressure. [556]
- In African-American hypertensive patients, vitamin D supplementation at a dose of 200,000 IU weekly for 3 weeks decreases systolic blood pressure by 9%. [557]
- The use of active vitamin D compounds in hypertensive patients is associated with a significant decrease in systolic blood pressure. [558]
- In patients with high blood pressure, oral vitamin D supplementation causes a reduction in systolic blood pressure. [559-560]
- In patients with high calcium levels, treatment of 1 µg alphacalcidol, a synthetic analogue of active vitamin D, for 6 months reduces diastolic blood pressure. [561-562]
- In patients with untreated mild hypertension, vitamin D treatment through short-term ultraviolet B exposure leads to lower blood pressure. [563-564]
- In elderly women, vitamin D3 supplementation improves blood pressure. [565]
- In men, vitamin D supplementation helps keep blood pressure within normal range. [566]
- In patients with vitamin D deficiency, weekly administration of 50,000 IU of oral vitamin D for 8 weeks as an adjunct supplement of antihypertensive drugs significantly reduces blood pressure. [567]
- In an elderly community-based population, intramuscular injections of 100,000 IU of vitamin D significantly lower blood pressure by improving arterial stiffness. [568]
- In dark-skinned patients, vitamin D supplementation appears to reduce systolic blood pressure. [569]
- A high dose of vitamin D3 (two capsules of 20,000IU/week) appears to have a more potent blood pressure-lowering effect. [570-573]
- In adults with vitamin D deficiency, high‐dose, 1‐year vitamin D supplementation appears to improve central blood pressure parameters. [574]
- Short-term, high-dose vitamin D administration for 1 year in hypertensive patients reduces blood pressure by improving blood vessel stiffness. [575-576]
- In healthy subjects and hypertensive patients, daily vitamin D3 therapy at a dose of >800 IU/day for <6 months significantly reduces systolic blood pressure. [577]
- In hypertensive subjects, vitamin D supplementation significantly lowers systolic blood pressure (an average of 6.1 mmHg) without any adverse side effects. [578-579]
- Vitamin D lowers blood pressure is by decreasing the concentrations of renin, aldosterone, angiotensin, and parathyroid hormone (PTH) levels– all of which constrict the blood vessels. [580-600]
J. Improves Cholesterol Levels
Just like hypertension and high blood sugar, elevated cholesterol levels can significantly increase one’s risk of developing fatal medical conditions. Fortunately, sun exposure and a high vitamin D diet can be a cost-effective way to bring down cholesterol levels within the normal range. In fact, several lines of evidence support the cholesterol-lowering effect of vitamin D:
- Low levels of vitamin D are strongly linked with abnormal cholesterol levels. [601-618]
- In professional rowers, vitamin D supplementation at a dose of 5,000 IU per day lowers blood cholesterol levels. [619]
- In school-aged patients, vitamin D supplementation increases the level of high-density lipoprotein (good cholesterol). [620]
- In postmenopausal women, vitamin D supplementation significantly decreases the level of low-density lipoprotein (bad cholesterol). [621]
- In statin-treated patients, vitamin D supplementation lowers cholesterol levels by improving its absorption. [622]
- In overweight patients, vitamin D supplementation significantly decreases LDL cholesterol. [623]
- In vitamin D-deficient men, increasing vitamin D concentrations through sunlight exposure significantly reduces triglyceride and LDL cholesterol levels. [624]
- Vitamin D receptors help increase cholesterol 7 alpha-hydroxylase, which in turn improves cholesterol metabolism. [625]
- In type 2 diabetic patients, administration of 2,000 IU daily of vitamin D3 for 18 months is associated with a significant decrease in total cholesterol and LDL cholesterol levels. [626]
- Vitamin D lowers cholesterol levels by improving the production of apolipoprotein A1. [627-628]
K. Helps Lose Weight
Vitamin D is also known as an anti-obesity nutrient, which is essential in maintaining a healthy weight. Several research studies found that the sunshine vitamin can help overweight and obese people lose weight safely and effectively:
- Lower levels of vitamin D are strongly linked with obesity and higher body mass index (BMI). [629-662]
- In overweight and obese women, 1000 IU of vitamin D daily significantly reduces body fat mass. [663]
- In overweight and obese women, 200 IU of vitamin D daily for 15 weeks significantly reduces fat mass and waist circumference. [664]
- Research suggests that vitamin D inhibits key molecules needed for the production of fat cells (adipocytes). [665-675]
- In overweight subjects, vitamin D supplementation improves weight loss without any adverse side effects. [676]
- In postmenopausal women, 2000 IU per day of oral vitamin D3 is associated with 5% to 10% reduction in body weight. [677]
- In healthy overweight and obese women, vitamin D supplementation significantly decreases body fat mass. [678]
- In obese children, vitamin D supplementation at a dose of 1200 IU for 26 weeks is associated with a significant reduction in BMI and body composition. [679]
- In healthy subjects with obesity, vitamin D supplementation at 25,000 IU per week or for 3 months significantly decreases body weight. [680]
- In older women, vitamin D supplementation augments the effect of weight loss. [681]
- In overweight and obese individuals on a weight-reducing program, vitamin D supplementation improves outcome measures. [682]
- In obese patients, long-term vitamin D supplementation enhances weight reduction. [683]
L. Improves Sleep Quality
Vitamin D levels are directly related to sleep quantity and quality. Therefore, boosting vitamin D levels through sun exposure, diet, and supplements may have beneficial effects on sleep. In fact, several high-quality studies suggest that vitamin D has positive effects on age-related sleeping difficulties as well as sleeping disorders:
- Vitamin D deficiency is strongly linked with poor sleep quality and various sleeping disorders. [684-699]
- A large body of preclinical studies found that vitamin D receptors are present in specific areas of the brain that are involved in sleep regulation. [700-704]
- In patients with sleeping problems, higher levels of supplemental vitamin D are associated with significant improvement in sleep quality. [705]
- In 20-50-year-old people with sleep disorders, vitamin D supplementation improves sleep quality, reduces sleep latency, raises sleep duration, and improves subjective sleep quality. [706]
- In veterans with multiple areas of chronic pain, standardized vitamin D supplementation improves sleep and quality of life by reducing pain levels. [707]
- In symptomatic menopausal women with sleep alterations, vitamin D supplementation (1 tablet per day for 24 weeks) improves sleep by reducing menopausal symptoms. [708]
M. Keeps Kidneys Healthy
There is a fascinating relationship between vitamin D and the kidneys. Whether from supplements or from the sun, the kidneys help convert vitamin D into its active form so that it can be effectively used by the body. Newer research shows that vitamin D may play a protective role against kidney disorders and keeping the kidneys working at optimum levels:
- Patients with chronic kidney disease (CKD) have abnormally low levels of vitamin D, suggesting that vitamin D replacement therapy may have a beneficial effect. [709-716]
- In patients with CKD, vitamin D replacement therapy prevents complications by decreasing excess levels of parathyroid hormone (which causes cardiovascular and bone complications). [717-728]
- In patients with CKD who are on dialysis, vitamin D therapy improves survival rate. [729-740]
- In animal models of glomerulonephritis (acute inflammation of the kidney), vitamin D therapy slows the progression of kidney disease. [741-747]
- In patients with CKD, supplementation with vitamin D is recommended at the early stage of the disease. [748]
- In patients with CKD and those undergoing hemodialysis, activated vitamin D treatment reduces all-cause and cardiovascular mortality rates. [749-752]
- In patients with CKD, vitamin D supplementation improves glomerular filtration rate, a measure of kidney function. [753-754]
- In patients on dialysis, vitamin D supplementation lowers the rate of kidney complications by preventing calcium buildup in the blood vessels. [755-764]
- In patients with CKD, vitamin D supplementation prevents the leakage of protein into the urine. [765-767]
- In animal models, vitamin D prevents kidney enlargement and scarring. [768]
- In hemodialysis patients, vitamin D supplementation improves kidney function by reducing the levels of proinflammatory cytokines, IL-8, IL-6, and TNF. [769]
- In patients with CKD, vitamin D supplementation improves blood flow to the kidney. [770]
N. Improves Sexual Function
With advancing age, sexual dysfunction occurs, resulting in decreased self-confidence and quality of life. Fortunately, vitamin D supplementation is one of the most cost-effective ways of ramping up sexual power in both men and women. There is accumulating evidence that vitamin D can help increase libido and restore sexual function while improving overall health:
- Low vitamin D status is strongly linked with low libido in both men and women, the severity of which depends on the degree of vitamin D deficiency. [771-772]
- In men, vitamin D deficiency contributes to erectile dysfunction. [773-774]
- Vitamin D may help improve erectile dysfunction by inhibiting inflammation and oxidative stress. [775-783]
- In young women with low vitamin D status, vitamin D treatment improves sexual desire, orgasm, and sexual satisfaction. [784]
- In men aged 20-49 years, vitamin D supplementation improves sexual function by increasing the levels of testosterone, the key male sex hormone that regulates libido. [785-786]
- Vitamin D can cause harder penile erections by increasing the levels of nitric oxide. [787-789]
O. Improves Fertility
Vitamin D also plays an integral part in reproduction. In women, vitamin D supplementation can drastically increase pregnancy success rate. In men, loading up on vitamin D has beneficial effects on sperm quality and quantity. A growing body of clinical evidence supports the many benefits of vitamin D on various fertility markers:
- Several high-quality studies suggest that vitamin D deficiency is strongly linked with infertility in both men and women. [790-810]
- Vitamin D deficiency also increases the risk of serious pregnancy complications, such as preeclampsia (characterized by hypertension, protein in the urine, and swelling), miscarriage, and preterm birth. [811-812]
- In women with polycystic ovary syndrome (PCOS), vitamin D supplementation normalizes ovulation by increasing sensitivity to follicle-stimulating hormone, which promotes the formation of egg or sperm. [813]
- In women with vitamin D deficiency, supplementation improves the ovarian reserve and delays the onset of menopause. [814]
- Patients with high initial vitamin D levels have a better chance of successful in vitro fertilization (IVF) compared to those with low vitamin D levels. [815-820]
- Vitamin D supplementation during pregnancy is associated with increased mean birth weight and reduced risk of small for gestational age birth. [821-822]
- Vitamin D supplementation during pregnancy improves maternal and fetal outcomes. [823-824]
- In pregnant women, vitamin D supplementation helps maintain a healthy pregnancy by reducing the risk of preeclampsia. [825]
- Vitamin D improves female fertility by aiding in the development and implantation of the uterus. [826-833]
- Vitamin D supplementation in men is associated with increased sperm survival. [834-835]
- In men, vitamin D supplementation improves sperm motility (the ability of sperm to move properly through the female reproductive tract to reach the egg). [836]
- In men with low sperm count, vitamin D supplementation significantly increases the chance of getting their partners pregnant by improving semen quality and quantity.[837-840]
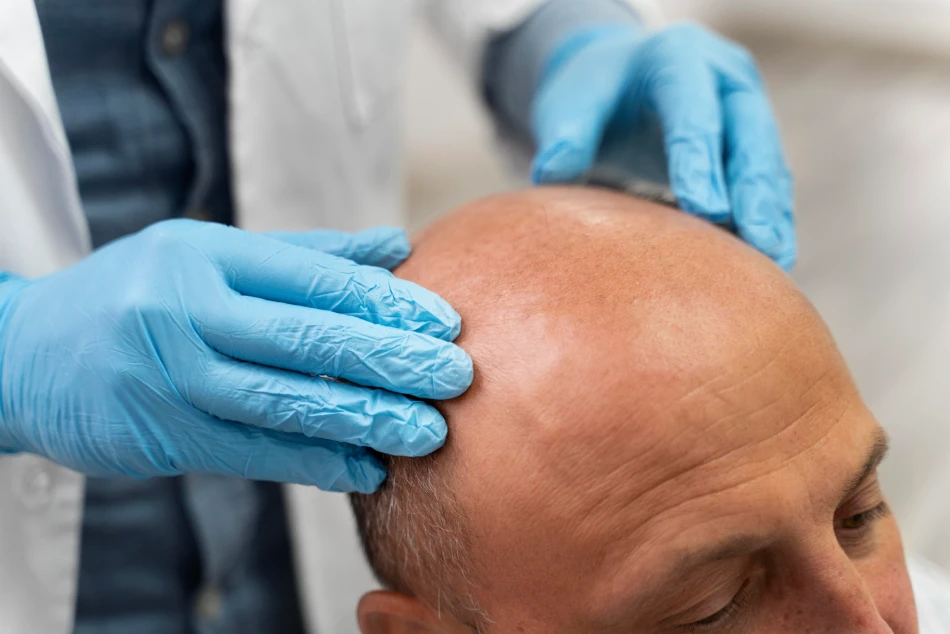
P. Prevents Hair Loss
Hair loss is inevitable and occurs with advancing age or it can be caused by other medical conditions. Aside from having a negative physical impact, hair loss often comes with devastating psychological consequences. Recently, medical professionals found a long-term, cost-effective solution to hair loss, thus eliminating the need for expensive medications and hair transplantation. Studies show that vitamin D supplementation alone can help fight hair loss:
- Poor vitamin D intake and low blood levels of vitamin D are associated with a higher prevalence of hair loss in both men and women. [841-860]
- Vitamin D is crucial for hair follicle integrity. [861-863]
- In patients with a single bald patch, three months of topical vitamin D therapy (applied on the scalp) is associated with better hair regrowth. [864-865]
- Vitamin D supplementation is effective in treating chemotherapy-induced hair loss. [866-868]
- Treatment with vitamin D analogs is associated with natural hair follicle formation and increased production of keratin, a protein that forms the main structural constituent of hair. [869-872]
- Nutritional supplementation of vitamin D or topical vitamin D analogues improves hair loss in patients with alopecia areata (a disease that causes hair to fall out in small patches). [873]
Vitamin D Supplementation
Vitamin D supplementation is a widely recommended practice to support overall health, particularly for individuals with limited sun exposure or dietary intake. Vitamin D plays a critical role in calcium absorption, which is essential for maintaining strong bones and preventing conditions like osteoporosis. It also supports immune system function, and muscle health, and may reduce the risk of certain chronic diseases, such as cardiovascular issues and autoimmune disorders.
Supplementing with vitamin D3 (cholecalciferol) is considered the most effective form for raising and maintaining optimal blood levels. The dosage varies depending on individual needs, but common daily doses range from 600 IU to 2000 IU for maintenance, with higher doses prescribed for treating deficiencies. Regular monitoring of blood levels can help ensure safe and effective supplementation while avoiding the risk of toxicity, which can occur with excessive intake.
For many, supplementation is an easy and affordable way to maintain adequate vitamin D levels, especially in winter months or regions with limited sunlight. While supplements can provide substantial benefits, combining them with moderate sun exposure and a diet rich in vitamin D sources, like fatty fish and fortified foods, offers a comprehensive approach to achieving and maintaining optimal health.
Vitamin D What does it do?
Vitamin D is a fat-soluble vitamin essential for maintaining strong bones and overall health. It plays a critical role in calcium and phosphorus absorption, which are necessary for bone mineralization and preventing conditions like osteoporosis and rickets. Without adequate vitamin D, bones can become thin, brittle, or misshapen, increasing the risk of fractures.
Beyond bone health, vitamin D supports immune system function by enhancing the body’s ability to fight infections and reduce inflammation. It influences cellular growth, neuromuscular function, and mood regulation, potentially lowering the risk of depression. Research also suggests that vitamin D may help prevent certain chronic diseases, including diabetes, cardiovascular conditions, and some types of cancer.
Vitamin D is unique because the body can produce it naturally when exposed to sunlight. However, factors like limited sun exposure, darker skin tones, and aging can lead to deficiencies. To ensure adequate levels, many people rely on dietary sources like fatty fish, fortified foods, or supplements, with vitamin D3 being the most effective form of supplementation.
What is Vitamin D Good for?
Vitamin D is essential for maintaining strong bones and teeth. It helps the body absorb calcium and phosphorus from the diet, which are critical for bone health and density. Without sufficient vitamin D, bones can become thin, brittle, or misshapen, leading to conditions like rickets in children or osteomalacia and osteoporosis in adults.
Beyond bone health, vitamin D supports the immune system, helping the body fight off infections and reduce inflammation. It plays a role in regulating immune responses, which is why adequate levels are associated with lower risks of autoimmune diseases and faster recovery from illnesses like the flu or respiratory infections.
Vitamin D also benefits mental and physical well-being. Research suggests it can help improve mood and reduce symptoms of depression, possibly by supporting serotonin production in the brain. Additionally, it contributes to muscle strength and coordination, reducing the risk of falls in older adults. Its overall impact extends to cardiovascular health, weight management, and even protection against certain chronic diseases.
Vitamin D Foods
Vitamin D is an essential nutrient found in various foods that help maintain bone health, immune function, and overall well-being. Fatty fish like salmon, mackerel, and sardines are among the richest natural sources, providing a significant amount of vitamin D. Cod liver oil is another potent option, offering a concentrated dose of the vitamin in a small serving.
Fortified foods also play a crucial role in boosting vitamin D intake, especially for individuals with limited sun exposure. Many dairy products, plant-based milk, orange juice, and breakfast cereals are enriched with vitamin D to help meet dietary needs. These foods are particularly beneficial for people who follow vegetarian or vegan diets, as they might not consume fish or other animal-derived sources.
For those looking to add variety to their diet, eggs and mushrooms offer smaller amounts of vitamin D. The vitamin is concentrated in the yolk of eggs, while certain mushrooms like maitake and UV-exposed portobellos provide a plant-based option. Including a mix of these foods in your meals can help maintain adequate vitamin D levels and support long-term health.
Vitamin D vs D3
Vitamin D is a vital nutrient that supports bone health, immune function, and overall well-being. It exists in two main forms: vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol). Both types help maintain adequate levels of calcium and phosphorus in the blood, but they differ in their sources and efficacy. Vitamin D2 is primarily derived from plant-based sources and fortified foods, while D3 comes from animal-based sources like fatty fish and eggs, as well as skin synthesis from sunlight exposure.
Vitamin D3 is generally considered more effective than D2 at raising and maintaining vitamin D levels in the body. Studies show that D3 is better absorbed and has a longer-lasting impact on blood levels of the vitamin. This makes it the preferred choice for supplementation, especially for individuals with a deficiency or those who have limited sun exposure.
When choosing between vitamin D and D3, D3 is often recommended due to its superior bioavailability and potency. However, D2 may still be a viable option for those following strict vegan or plant-based diets, as it is derived from non-animal sources. Ultimately, the choice should be based on dietary preferences, health needs, and professional medical advice.
Vitamin D Dosage
Vitamin D dosage varies based on age, health status, geographic location, and individual needs. For most adults, the recommended daily allowance (RDA) is 600 to 800 IU, which helps maintain adequate blood levels and supports bone health, immune function, and muscle strength. However, people with limited sun exposure or darker skin may require higher doses to achieve the same benefits. Supplementing with 1000 to 2000 IU daily is often safe and effective for maintaining optimal levels.
For individuals diagnosed with a vitamin D deficiency, higher doses are often prescribed. A common treatment plan involves taking 50,000 IU once a week for several weeks, followed by a maintenance dose. While high doses can correct deficiencies quickly, they should only be taken under medical supervision to avoid vitamin D toxicity, which can cause hypercalcemia and other complications. Regular blood tests are essential to monitor levels during high-dose supplementation.
Exceeding the tolerable upper intake level of 4000 IU daily without medical guidance can lead to health risks. For most, maintaining consistent supplementation with a moderate dose, combined with sun exposure and a vitamin D-rich diet, is sufficient to support overall health. Consulting with a healthcare provider is the best way to determine the appropriate vitamin D dosage tailored to individual needs.
Vitamin D Benefits for Men
Vitamin D offers numerous benefits for men, particularly in supporting bone health and muscle function. By enhancing calcium absorption, it strengthens bones and reduces the risk of fractures, which is especially important as men age and bone density naturally declines. Additionally, vitamin D helps improve muscle strength and coordination, reducing the risk of falls and injuries.
This essential nutrient also plays a critical role in boosting immune function and reducing inflammation. Men with sufficient vitamin D levels are less likely to experience severe infections and may have a lower risk of chronic conditions such as heart disease and diabetes. Furthermore, vitamin D supports healthy testosterone levels, which are vital for maintaining energy, libido, and overall vitality.
Mental health is another area where vitamin D proves beneficial for men. Research suggests that adequate levels can improve mood and reduce symptoms of depression. It may also help regulate stress levels and promote better sleep quality, contributing to overall mental and physical well-being. With these wide-ranging benefits, maintaining optimal vitamin D levels is essential for men of all ages.
Side Effects of Vitamin D 50000 IU
Taking 50,000 IU of vitamin D is typically prescribed for severe deficiencies, but it can cause side effects if used improperly. One of the most common issues is hypercalcemia, which occurs when calcium levels in the blood become too high. Symptoms of hypercalcemia include nausea, vomiting, confusion, and kidney problems, which may require medical attention.
Long-term use of high doses of vitamin D can also lead to kidney stones, as the excess calcium in the body may form deposits in the kidneys. This can cause pain, urinary problems, and damage to the kidneys over time. It’s important to monitor kidney function during treatment with such high doses.
In rare cases, excessive vitamin D intake may lead to toxicity, which can affect the heart and other organs. Symptoms of vitamin D toxicity include weakness, fatigue, dehydration, and loss of appetite. It’s essential to follow a healthcare provider’s guidance when taking high-dose vitamin D to avoid these adverse effects.
Low Vitamin D and Weight Gain
Low vitamin D levels have been linked to weight gain and difficulty losing weight. Research suggests that vitamin D plays a role in regulating fat metabolism, and a deficiency may disrupt this process. People with low vitamin D levels may experience an increase in fat storage, especially in the abdominal area, leading to weight gain. The hormone leptin, which regulates hunger and fat storage, can also be affected by insufficient vitamin D, potentially leading to overeating.
Additionally, low vitamin D is associated with reduced energy levels and muscle weakness. This can make physical activity feel more difficult or tiring, further contributing to weight gain or difficulty losing weight. A lack of sunlight exposure, which is the primary source of vitamin D, can exacerbate these effects, especially in colder months when people tend to be less active and spend more time indoors.
Improving vitamin D levels through supplementation, sunlight exposure, and vitamin D-rich foods may help support healthy weight management. Studies suggest that correcting a deficiency may boost metabolism, improve energy levels, and support better fat burning. However, more research is needed to fully understand the connection between vitamin D and weight gain.
Vitamin D3 Benefits Weight Loss
Vitamin D3 may play a role in weight loss by helping regulate fat metabolism. Studies suggest that individuals with adequate vitamin D levels are more likely to maintain a healthy weight, as the nutrient supports the body’s ability to metabolize fat effectively. Low vitamin D levels have been linked to an increased risk of obesity, possibly due to its influence on fat storage and energy expenditure.
Additionally, vitamin D3 may impact hormones that regulate hunger and satiety. It has been shown to help balance insulin levels, which can reduce cravings and improve blood sugar control. This, in turn, may lead to better weight management and prevent overeating. Some research also indicates that vitamin D deficiency may contribute to an imbalance in appetite-regulating hormones, making it harder to control calorie intake.
Moreover, vitamin D3’s role in reducing inflammation could indirectly support weight loss efforts. Chronic inflammation is often linked to obesity and metabolic disorders. By lowering inflammation, vitamin D3 may improve insulin sensitivity and fat burning, making it easier for the body to lose weight. However, more research is needed to fully understand how vitamin D3 influences weight loss.
Vitamin D Injection
A vitamin D injection is a medical treatment used to address significant vitamin D deficiencies, especially when oral supplementation is not effective or practical. This form of vitamin D, typically in the form of vitamin D2 or D3, is administered directly into the bloodstream, allowing for rapid absorption and a more immediate effect. Injections are often prescribed when a person’s vitamin D levels are dangerously low, and other methods, such as dietary changes or supplements, are not sufficient.
Vitamin D injections are often used in individuals with conditions that impair the absorption of nutrients, such as celiac disease or Crohn’s disease, or in people who are severely deficient and need to replenish their levels quickly. They can also be helpful for individuals who have difficulty absorbing oral supplements due to gastrointestinal issues or those who cannot tolerate high doses of oral vitamin D.
The effects of a vitamin D injection can last for several weeks to months, depending on the dose administered and the individual’s needs. Common side effects may include mild pain at the injection site, nausea, or dizziness. However, vitamin D injections are generally well-tolerated and are considered a safe and effective treatment option for those with severe deficiencies. As with any medical treatment, they should be administered under the supervision of a healthcare professional to ensure safety and proper dosage.
Vitamin D Normal Range
The normal range for vitamin D levels in the body typically falls between 30 and 50 ng/mL (nanograms per milliliter). This range is considered sufficient for most individuals to support optimal bone health, immune function, and overall well-being. Levels within this range indicate that a person has enough vitamin D to effectively absorb calcium and phosphorus and maintain healthy bones and teeth.
Levels below 20 ng/mL are generally considered deficient, which can lead to bone weakness, muscle pain, and increased susceptibility to infections. Deficiency can also result in conditions like osteomalacia in adults or rickets in children, both of which affect bone development. Individuals with low vitamin D levels may experience fatigue, depression, or muscle weakness.
While levels above 50 ng/mL are generally safe, extremely high levels (above 100 ng/mL) may indicate toxicity. This can lead to harmful effects, such as hypercalcemia (high calcium levels), which can damage organs and tissues. It is important to monitor vitamin D levels and consult a healthcare provider to ensure they remain within a healthy range, especially when taking supplements.
Vitamin D Tablets
Vitamin D tablets are a popular and convenient way to ensure adequate vitamin D intake, especially for individuals who may have limited sun exposure or dietary sources of this essential nutrient. They are available in various forms, with vitamin D3 (cholecalciferol) being the most commonly recommended due to its superior absorption and effectiveness in raising vitamin D levels in the body. These tablets come in different dosages, typically ranging from 600 IU to 5000 IU per tablet, depending on individual needs and health conditions.
Taking vitamin D tablets can be beneficial for supporting bone health, enhancing immune function, and improving mood. Vitamin D plays a crucial role in calcium absorption, which helps maintain strong bones and teeth, while also contributing to the proper functioning of the immune system. For those who suffer from a deficiency, supplementation with vitamin D tablets can help restore optimal levels and prevent complications like bone pain, fatigue, and increased susceptibility to infections.
While vitamin D supplements are generally safe, it is important to follow the recommended dosage and consult a healthcare provider before starting a regimen. Excessive intake of vitamin D can lead to toxicity, causing symptoms such as nausea, kidney stones, and hypercalcemia. Regular monitoring of vitamin D levels is essential to ensure that supplementation is both safe and effective.
How much Vitamin D Per Day?
The amount of vitamin D you need per day depends on factors such as age, health status, and exposure to sunlight. For most adults, the recommended daily allowance (RDA) is between 600 and 800 IU. Older adults, people with darker skin, or those who spend limited time in the sun may need more to maintain optimal levels.
If you have a deficiency, your doctor may recommend higher doses, such as 1,000–2,000 IU per day. Some people, especially those with certain medical conditions like osteoporosis, may need even higher doses. However, it’s essential to follow medical guidance to avoid excessive intake, which can lead to toxicity.
Generally, taking up to 4,000 IU daily is considered safe for most people. Doses above 10,000 IU daily should only be taken under medical supervision. Regular blood tests can help determine if you need to adjust your vitamin D intake to ensure you are meeting your specific health needs.