Overall Health Benefits of Oxytocin
Oxytocin offers broad health benefits, including anti-aging effects, weight management, stress reduction, mood enhancement, cognitive and social skill improvements, blood pressure regulation, cardiovascular protection, inflammation reduction, addiction treatment, sexual function enhancement, blood sugar control, and better sleep quality.
- Produces anti-aging effects [1-11]
- Promotes weight loss [12-36]
- Fights stress [37-50]
- Improves mood [51-64]
- Improves cognitive function [65-79]
- Improves social skills [80-98]
- Lowers blood pressure [99-107]
- Lowers the risk of cardiovascular disease [108-123]
- Fights inflammation [122, 124-135]
- Treats substance addiction [136-161]
- Improves sexual function [162-213]
- Improves blood sugar levels [214-223]
- Improves sleep quality [224-231]
Key Takeaways
- Anti-Aging and Longevity: Oxytocin has anti-aging properties, helping to slow cellular aging and promote overall vitality.
Mood and Stress Regulation: Known as the “love hormone,” oxytocin helps alleviate stress, improves mood, and enhances social interactions, making it beneficial for mental well-being. - Weight Management and Metabolism: It supports weight loss and metabolic health, contributing to better body composition and blood sugar control.
- Cardiovascular Health: Oxytocin lowers blood pressure and reduces cardiovascular risk, protecting heart health over time.
- Enhanced Cognitive and Sexual Function: This hormone improves cognitive function, memory, and social skills, while also enhancing sexual satisfaction and intimacy.
What is Oxytocin?
Oxytocin is a nano peptide hormone that is produced by the posterior pituitary gland. In women, this hormone is released in large amounts during the process of childbirth to stimulate the uterine muscles to contract. Other factors such as nipple stimulation and breastfeeding can also increase the secretion of oxytocin in women. Both men and women also release the hormone during skin- to-skin contact, sexual arousal, and orgasm/ejaculation. Moreover, oxytocin plays a major role in different human behaviors such as trust, bonding, desire, and social recognition. Because of this, oxytocin is called the “cuddle” or “love” hormone.
How Oxytocin Works
IMG
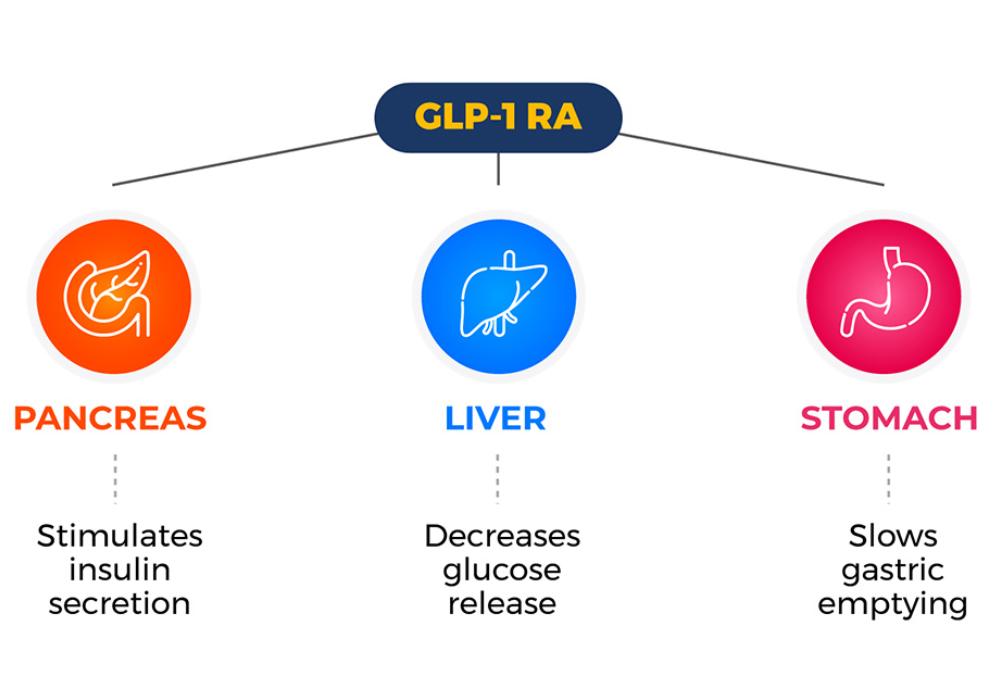
Oxytocin is produced in the hypothalamus. The posterior pituitary gland then secretes the hormone into the bloodstream. Oxytocin travels to the uterus and increases both the intensity and frequency of contractions. This in turn speeds up the delivery of the baby and the placenta and reduces the risk of heavy bleeding. In men, oxytocin is also present and plays a role in sperm transport and testosterone production by the testes. The health benefits of oxytocin go beyond these mechanisms. For instance, oxytocin can help promote weight loss by stimulating the breakdown of fat. It can also help keep blood sugar within normal limits by boosting the body’s response to the hormone insulin (insulin sensitivity).
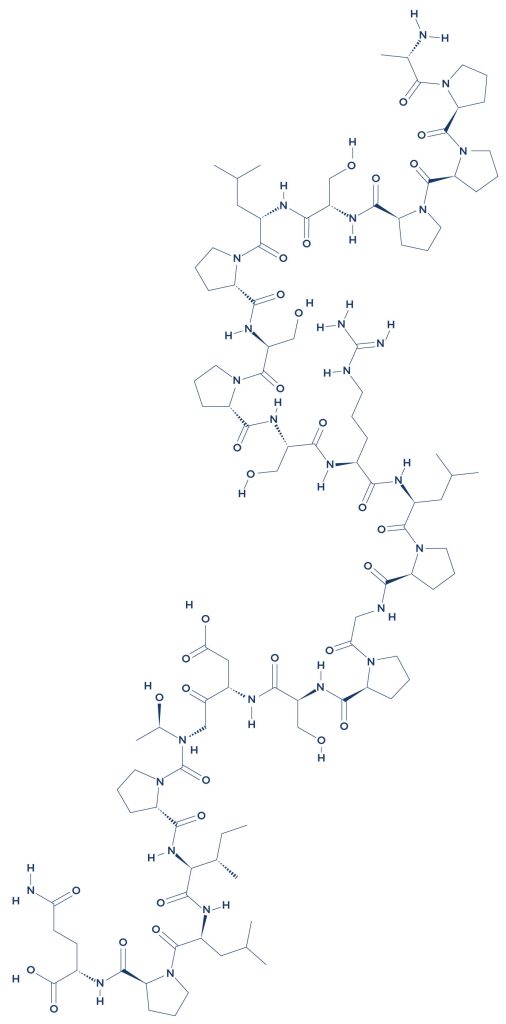
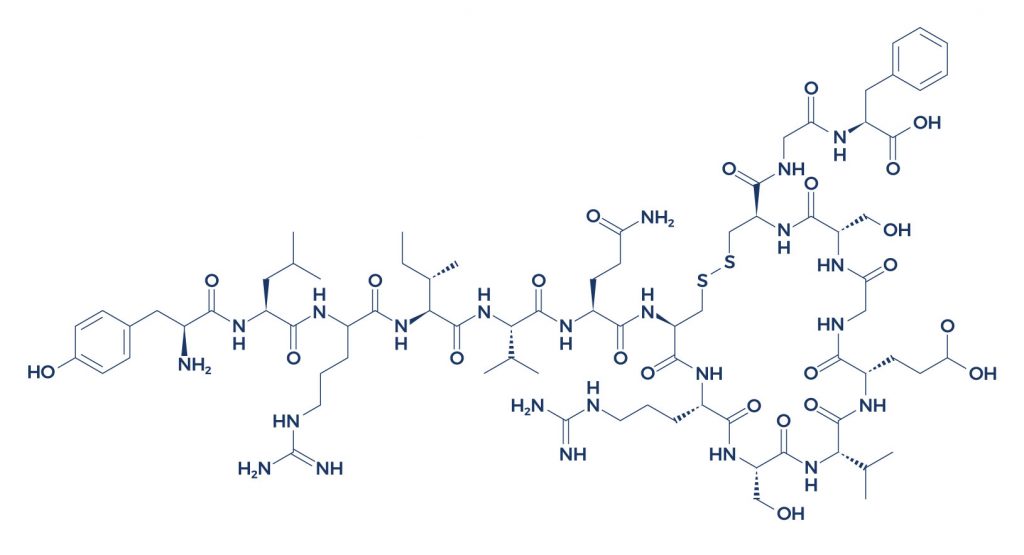
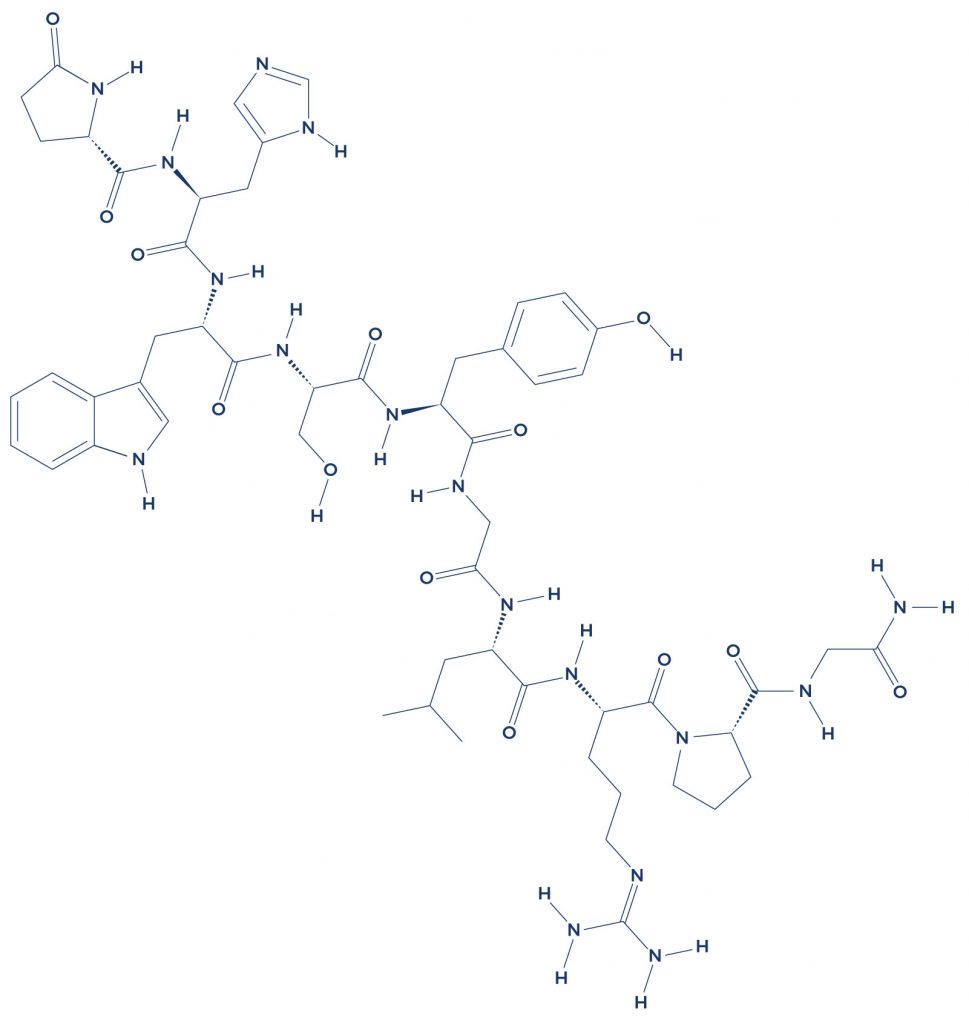
Chemical Structure of Oxytocin
IMG
Research on Oxytocin
A. Produces Anti-Aging Effects

Oxytocin can help slow down the signs of aging by preventing the release of proinflammatory cytokines, which may speed up the process of skin aging via inhibition of collagen synthesis and enhanced collagen degradation. [1] Since collagen is responsible for maintaining the structures of the skin, decreased collagen levels may contribute to the formation of fine lines, wrinkles, and other age-related spots. Oxytocin can also help alleviate age-related changes in cell structures.
A number of convincing evidence support the anti-aging effects of oxytocin:
- In human skin cells, treatment with oxytocin for 12 days prevented the accumulation of senescent keratinocytes and fibroblasts which are associated with age-related loss of skin function and integrity. [2]
- In female subjects aged 48-61 years, higher oxytocin levels were associated with a lower skin age score (SAS), suggesting a more youthful appearing skin. [3]
- In female prairie voles, daily oxytocin injections prevented cellular aging caused by social isolation. [4-5]
- In old mice, the administration of oxytocin rapidly improved muscle regeneration by enhancing aged muscle stem cell activation via activation of the MAPK/ERK signalling pathway. [6]
- In elderly mice, oxytocin promoted liver regeneration as evidenced by an increase in the number of liver cells called hepatocytes which decrease with aging. [7]
- Oxytocin has been shown to increase lifespan by increasing the length of telomeres which are repeated sequences of non-coding DNA located at the terminal ends of chromosomes that play an integral role in chromosome stability and longevity. [8]
- Sudden bursts of oxytocin have been found to suppress inflammaging, which is the age-related increase in inflammation. [9]
- In male and female rats, oxytocin-mediated social enrichment promoted longer telomeres which are associated with longevity and stable chromosomes. [10]
- In older mice, the administration of oxytocin rapidly improved muscle regeneration by enhancing aged muscle stem cell activation/proliferation. [11]
B. Promotes Weight Loss

Oxytocin has the ability to promote weight loss by reducing food consumption and increasing energy expenditure. [12] It does this by affecting certain chemicals in the brain that increase the feelings of fullness or satiety. With increased energy expenditure, no additional fat will be stored in the body. Instead, the stored fats will be burned as a source of energy for various cellular activities.
Evidence suggests that oxytocin administration can help increase lean muscle mass and reduce body fat percentage:
- In obese, older adults, intranasal oxytocin administration resulted in a significant increase of 2.25 kg in whole-body lean mass without any adverse events compared with placebo. [13]
- In diet-induced obese (DIO) and genetically obese mice and rats, oxytocin administration resulted in weight loss by reducing energy intake and increasing fatty tissue breakdown. [14-26]
- In obese men, oxytocin markedly reduced hunger-driven food intake in the fasted state and led to a reduction in snack consumption compared to men with normal weight. [27]
- A review of multiple studies found that 8 weeks of oxytocin treatment in adults with obesity or overweight led to substantial weight loss. [28]
- In obese women of childbearing age, oxytocin deficiency was common compared with non-obese women. [29]
- In obese monkeys, oxytocin administration for 2 weeks inhibited food intake, increased energy expenditure, and produced weight loss. [30]
- In healthy men, intranasal oxytocin reduced caloric intake without any adverse side effects. [31]
- Several studies suggest that oxytocin can be a potential treatment for obesity and related metabolic diseases. [32-35]
- In healthy women, the administration of 24 IU of intranasal oxytocin specifically reduced food cravings in the cognitive control condition compared with placebo. [36]
C. Fights Stress

The stress response consists of a series of events in the brain that stimulate the hypothalamic–pituitary–adrenal (HPA) axis. When activated, the HPA axis stimulates the release of corticotropin-releasing factor (CRF), a peptide hormone involved in the stress response. CRF promotes the production and secretion of glucocorticoids in response to stress. Oxytocin has been shown to fight stress by inhibiting ACTH and subsequent glucocorticoid secretion. [37-38]
A number of studies support the anti-stress properties of oxytocin:
- In healthy men exposed to the Trier Social Stress Test, the combination of oxytocin and social support significantly reduced the levels of the stress hormone cortisol as well as anxiety and increased calmness during stress. [39]
- In female rats given central infusions of oxytocin, a decrease in anxiety-related behaviors was observed. [40]
- In healthy male and female participants who were subjected to a standardized laboratory stressor, the Trier Social Stress Test, oxytocin secretion exerted a protective function against the health-compromising effects of sustained stress. [41]
- A study found that oxytocin release is associated with self-soothing behaviors that can help reduce stress. [42]
- In healthy male and female participants, oxytocin reduced stress-evoked responses in the amygdala, the brain region associated with emotional processes. [43]
- In healthy men exposed to social stress, intranasal administration of oxytocin (24 IU) reduced perceived social stress. [44]
- In monkeys, intranasal oxytocin administration attenuated the ACTH stress response. [45]
- In breastfeeding mothers, oxytocin release is associated with a reduction in stressful behaviors. [46]
- Intranasal oxytocin administration was shown to modulate the activity of the amygdala which in turn reduces fear, anger, and feelings of mistrust. [47-50]
D. Improves Mood

As one of the feel-good hormones, oxytocin can produce a calming effect which can positively affect the overall mood. By decreasing the levels of the stress hormone cortisol, oxytocin may produce anti-anxiety and antidepressant effects. [51]
The mood-boosting effects of oxytocin are backed by a number of studies:
- In male rats, oxytocin administration at doses of 0.01 and 0.1 μg/kg subcutaneously (SC) reduced background anxiety but not when delivered into the cerebroventricular system (ventricles of the brain). [52]
- Intracerebroventricular administration of oxytocin in male rats produced anti-anxiety effects by activating the extracellular signal-regulated kinase 1/2 (ERK1/2) cascade in the brain which is involved in a wide array of cellular functions. [53]
- In patients with generalized social anxiety disorder (GSAD), acute intranasal administration of oxytocin produced anti-anxiety effects as evidenced by decreased amygdala reactivity to fearful faces. [54]
- In male Wistar rats with autism spectrum disorder (ASD), the administration of oxytocin via the brain reduced the anxiety to the healthy control level. [55]
- In athletes, winners had significantly lower cognitive anxiety and higher self-confidence scores and oxytocin levels than losers. [56]
- Youth with a separation anxiety disorder had significantly lower salivary oxytocin levels than clinically anxious youth without the disorder. [57]
- In mice, long-term isolation reduced the levels of oxytocin in the brain which contributed to the development of depression and anxiety-related behaviors. [58]
- In high-trait anxious males, oxytocin attenuated negative cognitive responses to stress. [59]
- In men, oxytocin decreased the stress of negative social interactions. [60]
- In rats, oxytocin treatment decreased anxiety-like behaviors by lowering cortisol levels. [61]
- A review of multiple studies concluded that oxytocin can be a promising therapeutic option for human anxiety disorders especially those associated with socio-emotional dysfunctions. [62]
- In adult male and female rats, oxytocin administration prevented the increase in behavioral despair due to maternal separation. [63]
- In postpartum mothers, oxytocin administration reduced the risk of postpartum depression. [64]
E. Improves Cognitive Function
Oxytocin is necessary for optimum brain function since brain regions that are crucial for higher cognitive functions such as the prefrontal cortex and the hippocampus have large amounts of oxytocin receptors. [65-66] In addition, oxytocin has also been found to enhance the transmission of nerve signals necessary for vital cognitive processes such as learning and memory. [67]
There’s a wealth of evidence showing that oxytocin has a significant effect on various cognitive abilities:
- In mice lacking the oxytocin gene, significant deficits in learning and memory were observed. [68-71]
- In human subjects, intranasal oxytocin administration improved memory for facial recognition. [72-74]
- In healthy human male volunteers who received oxytocin, an improvement in memory recall for previously seen happy faces compared with angry and neutral human faces was observed. [75]
- In healthy male volunteers, intranasal administration of 24 IU oxytocin improved performance on the Reading the Mind in the Eyes Test (RMET). [76]
- In human volunteers, intranasal oxytocin administration improved memory recall for words compared with placebo. [77]
- In overweight and obese men, the administration of 24 IU single-dose intranasal oxytocin improved reaction times in the choice-reaction task (go task) and displayed fewer stop errors (participants had to withhold their response when prompted). [78]
- In female rats with Alzheimer’s disease, intranasal oxytocin restored cognitive function by suppressing the formation of abnormal protein structures such as beta- amyloid and tau tangles. [79]
F. Improves Social Skills
Oxytocin plays an integral role in social bonding. Also known as the “love hormone”, it is involved in the regulation of emotions and acts as a chemical reward in the brain during various social interactions. Since many oxytocin receptors are distributed in various brain regions, this hormone may play a role in regulating social behaviors. [80-81]
Studies show that oxytocin can help address impaired social skills due to various medical conditions:
- In patients with autism, intranasal administration of oxytocin resulted in stronger interactions with a partner during a simulated ball game and enhanced feelings of trust and preference. [82]
- In a 16-year-old girl with autistic disorder, long-term administration of oxytocin nasal spray improved social interactions and social communication without adverse effects and dramatically decreased aberrant behaviors such as irritability and aggressiveness. [83]
- In patients with social anxiety disorder (SAD), intranasal administration of the hormone oxytocin (24 IU) improved observer-rated social behavior compared with placebo. [84]
- In patients with autism spectrum disorder (ASD), treatment with intranasal oxytocin for 6 weeks resulted in improvement in emotion recognition. [85]
- In children with autism aged 6-12 years, 4-week intranasal oxytocin treatment (24 International Units, twice daily) significantly enhanced social abilities compared with placebo. [86]
- In older males, the administration of oxytocin nasal spray (20 international units) improved emotion recognition compared with placebo. [87]
- In women with post-traumatic stress disorder (PTSD), a single intranasal dose of oxytocin enhanced compassion toward women. [88]
- In participants who self-administered one dose of 24 IU of oxytocin, a positive effect on the accuracy of fear recognition was observed. [89]
- In patients with schizophrenia, a mental disorder that affects the interpretation of reality, intranasal oxytocin administration resulted in improvements in fear recognition, perspective taking, and a reduction in negative symptoms. [90]
- In adult males with and without autism, intranasal administration of 24 IU of oxytocin enhanced gaze to the eyes. [91]
- In newborn monkeys, oxytocin administration increased facial gesturing at a human caregiver compared with placebo. [92]
- Reviews of multiple studies found that the administration of oxytocin to both individuals with ASD and typically developing individuals can enhance performance on social cognitive tasks. [93-95]
- In infants with Prader-Willi syndrome, a rare genetic disorder that causes physical, mental, and behavioral problems, the administration of 4 IU of oxytocin either every other day, daily, or twice daily resulted in significant improvements in Clinical Global Impression scale scores, social withdrawal behavior, and mother-infant interactions. [96]
- In children with autism, oxytocin administration improved brain function as evidenced by enhanced social functioning. [97]
- A study found that oxytocin modulates human communication by enhancing cognitive exploration. [98]
G. Lowers Blood Pressure
Oxytocin may exert beneficial effects in elevated blood pressure or hypertension. Several mechanisms associated with the blood pressure-lowering effects of oxytocin include dilation of the blood vessels, excretion of the electrolyte called sodium, and increased urine excretion. [99]
Evidence suggests that oxytocin administration may help lower blood pressure in people with hypertension:
- In women with severe preeclampsia, a complication of pregnancy characterized by hypertension, protein in the urine, and swelling of the legs, an intravenous bolus of 5 IU oxytocin resulted in a decrease in systemic vascular resistance and blood pressure. [100]
- In rats, oxytocin prevented hypertension due to low oxygen levels. [101]
- In rats with hypertension caused by exposure to loud noise, oxytocin administration decreased blood pressure after 24 hours. [102]
- The administration of oxytocin at a dose of 1 mg/kg subcutaneously in hypertensive rats enhanced the effects of the blood pressure-lowering medication clonidine. [103]
- In rats that had surgical removal of the ovaries, once daily oxytocin injection for 12 days decreased blood pressure for 3 weeks. [104]
- In male and female rats, oxytocin administration resulted in a long-term reduction in blood pressure. [105-106]
- In male rats, a significant decrease in blood pressure was observed after oxytocin administration compared to placebo treatment. [107]
H. Lowers the Risk of Cardiovascular Disease
Oxytocin may help protect against cardiovascular disease. It exerts cardioprotective effects by promoting the formation of new blood vessels in the heart, reducing proinflammatory cytokines and immune cell infiltration, decreasing oxidative stress, preventing heart muscle enlargement, and increasing the viability of heart cells (cardiomyocytes). [108]
A compelling number of evidence suggests that oxytocin may significantly lower the risk of cardiovascular disease:
- In human heart cells, oxytocin promoted cell activation and heart regeneration after cardiac injury. [109]
- In anesthetized rats, intraperitoneal administration of oxytocin reduced the size of dead heart tissue. [110-112]
- In female rats, oxytocin administration improved heart rate and force of heart contraction. [113]
- In rats that received oxytocin, a significant decrease in heart injury due to insufficient blood flow was observed. [114]
- Studies suggest that cardiac injury is accelerated by a deficit of oxytocin receptors in the heart. [115-116]
- In mice, oxytocin attenuated atherosclerosis (plaque formation in the heart arteries) and fat tissue inflammation. [117]
- In adult rats, oxytocin induced regeneration of heart cells via activation of the cardiac oxytocin system. [118]
- A study reported that oxytocin can attenuate atherosclerosis by decreasing superoxide production and release of proinflammatory cytokines. [119]
- In neonatal rat cardiomyocytes, oxytocin increased glucose uptake which may play a major role in the maintenance of cardiac function and cell survival during metabolic stress. [120]
- In a rabbit model of myocardial infarction, oxytocin improved cardiac function by activating cell-survival signals and angiogenesis (blood vessel formation). [121]
- In a rat model of myocardial infarction, oxytocin improved the function of the injured heart by reducing inflammation and programmed cell death (apoptosis). [122-123]
I. Fights Inflammation
Oxytocin may be beneficial in the treatment of various inflammatory conditions. This hormone can ward off these conditions by suppressing the production of inflammatory substances and signaling pathways involved in the body’s natural inflammatory response.
The anti-inflammatory properties of oxytocin are backed by a number of studies:
- In a rat model of myocardial infarction, oxytocin reduced inflammation and programmed cell death (apoptosis). [122]
- In mice with lipopolysaccharide (LPS)-induced lung injury, oxytocin reduced the associated lung damage and the levels of the inflammatory substances interleukin (IL)-1β, IL-18, and IL-6. [124-126]
- In a rat model of seizure disorder, oxytocin treatment reduced neuroinflammation and seizure activity. [127]
- In healthy men, oxytocin treatment reduced the levels of inflammatory cytokines caused by bacterial endotoxin. [128]
- Recent studies reported that oxytocin may shorten the recovery period of COVID-19 patients via modulation of inflammatory, immune, and restorative functions of the immune system. [129-130]
- In a rat model of lung injury due to blood infection (sepsis), oxytocin injection decreased the levels of inflammatory substances such as plasma malondialdehyde (MDA), lactic acid (LA), C-reactive protein (CRP), interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and interleukin 1-beta (IL 1-β). [131]
- In rats, oxytocin administration attenuated increased sensitivity to pain and inflammation. [132]
- In mice, intranasal administration of oxytocin reduced the subsequent release of pro-inflammatory factors. [133]
- Subcutaneous administration of oxytocin at 100 and 1000 micrograms per kilogram reduced carrageenan-induced inflammation in rat hindpaw. [134]
- In female Sprague-Dawley rats, oxytocin protected against sepsis-induced oxidative damage and exerted anti-inflammatory effects via inhibition of neutrophil infiltration. [135]
J. Treats Substance Addiction
Oxytocin plays an integral role in reward, stress, social affiliation, and various cognitive functions. Because of this, there is increasing interest in this hormone as a potential therapeutic option for substance addiction. There are several mechanisms that can help reduce substance cravings. Oxytocin does this by increasing the levels of brain chemicals (neurotransmitters) such as dopamine and serotonin. [136-137] Dopamine regulates the brain’s pleasure and reward systems while serotonin regulates appetite, mood, and body temperature.
An increasing number of studies support the beneficial effects of oxytocin on substance addiction:
- In several animal models of addiction, oxytocin was found to decrease the self-administration of a number of addictive substances. [138-146]
- A study reported that oxytocin may reduce addictive behaviors by restoring abnormal drug-induced changes in the glutamatergic system which contains the major excitatory neurotransmitter in the nervous system called glutamate. [147]
- In male and female rats with cocaine addiction, oxytocin decreased reinstated cocaine-seeking behaviors. [148]
- In alcohol-dependent rats and humans, intranasal oxytocin (24 IU) decreased alcohol cravings. [149]
- In mice, oxytocin (0, 0.3, 1, 3, or 10 mg/kg) dose-dependently reduced ethanol consumption. [150]
- In subjects with alcohol abuse, oxytocin treatment significantly improved social perception and reduced cue-induced alcohol cravings and appetitive approach bias. [151]
- In heroin-dependent patients, oxytocin administration reduced cravings and withdrawal symptoms. [152]
- In rat models of methamphetamine addiction, oxytocin decreased methamphetamine self-administration, methamphetamine hyperactivity, and relapse to methamphetamine-seeking behaviors. [153-157]
- In methamphetamine-dependent patients undergoing stimulant abuse treatment, intranasal oxytocin administration for 4 weeks at 40 IU resulted in a significant improvement in the craving and depression scores compared with normal saline. [158]
- In a rat model of alcohol dependence, oxytocin specifically and selectively blocked the enhanced motivation for alcohol drinking. [159]
- In a patient with alcohol dependence and oropharyngeal cancer, oxytocin administration decreased the severity of addiction and led to an improvement in the quality of life. [160]
- A review of multiple studies reported that oxytocin administration in opioid-dependent individuals decreased tolerance to the drug and improved withdrawal symptoms. [161]
K. Improves Sexual Function
Also known as the “love hormone”, oxytocin increases bonding and sexual intimacy. The more oxytocin the body produces, the more physical intimacy is experienced. In addition, oxytocin has been found to facilitate penile erection in men by increasing the levels of nitric oxide, a substance that widens the blood vessels to increase penile blood flow. [162] In women, oxytocin can help relieve unpleasant age-related symptoms such as vaginal dryness and painful sexual intercourse. [163]
A good deal of evidence suggests that oxytocin administration can help improve sexual function in men and women experiencing sexual health issues due to aging and other medical conditions:
- In a male patient with treatment-resistant anorgasmia (delayed, infrequent, or absent orgasms), the administration of intranasal oxytocin before sexual intercourse restored ejaculation. [164]
- In male rats, systemic oxytocin administration significantly shortened the latency to the first mount, intromission (the penis enters the vagina during a mount), and ejaculation during copulatory behavior. [165-166]
- In premenopausal and postmenopausal women, long-term intranasal oxytocin administration improved sexual function and symptoms of depression. [167]
- In postmenopausal women with vaginal atrophy (thinning, drying, and inflammation of the vaginal walls) and sexual dysfunction, the administration of an oxytocin vaginal gel improved all domains of sexual function such as desire, arousal, lubrication, pain, and sexual satisfaction. [168-169]
- In a patient with sexual dysfunction due to anxiety, oxytocin administration positively impacted a number of components of sexual function such as libido, erection, and orgasm with no adverse side effects. [170]
- Intranasal oxytocin administration was associated with higher levels of sexual satiety after sexual intercourse in men and better abilities to share sexual desires or empathize with a partner in women. [171-172]
- Several male rat studies showed that the injection of oxytocin into specific brain regions can induce penile erection and that the administration of substances that counters the effects of oxytocin can prevent oxytocin-induced penile erection. [173-201]
- In female rats, the administration of oxytocin is associated with increased sexual behaviors such as lordosis (a reflexive posture in response to male sexual stimulation to facilitate intromission) and receptivity (female responses necessary for a successful intravaginal ejaculation). [202-209]
- In male rabbits, an infusion of oxytocin resulted in increased latencies to the first mount, intromission, and post-ejaculatory refractory periods. [210-211]
- In female hamsters, oxytocin injection induced sexual receptivity in a dose-dependent manner. [212]
- Female treatment with intranasal oxytocin improved male sexual quality of life and evaluation of female partner’s sexual performance. [213]
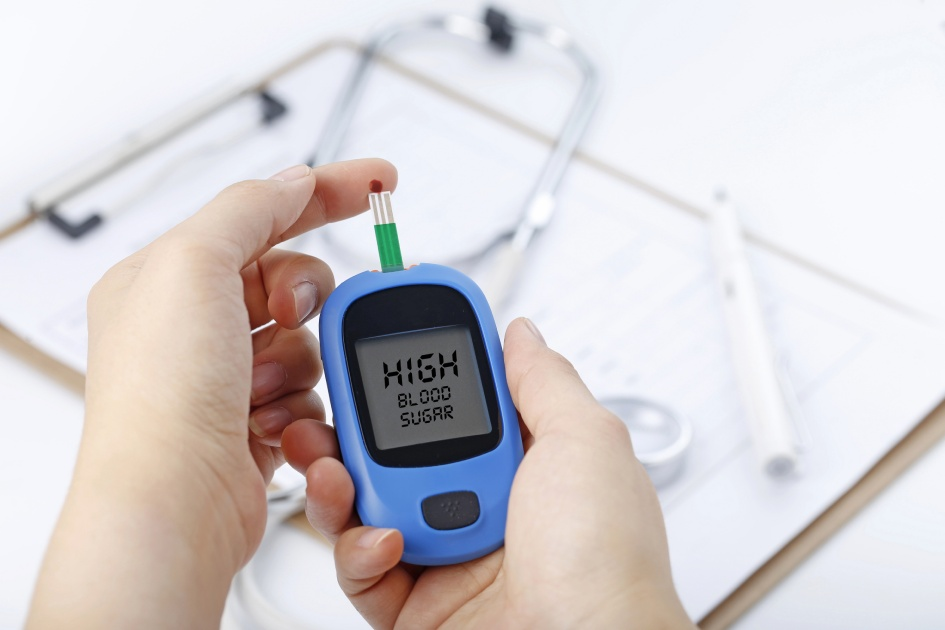
L. Improves Blood Sugar Levels
Oxytocin can help keep blood sugar within normal limits by boosting the body’s response to the hormone insulin known as insulin sensitivity. Another mechanism is that oxytocin increases the rate of blood sugar uptake, which allows cells to utilize blood sugar effectively for various important biological functions. [214]
Studies show that oxytocin administration can help improve blood sugar levels:
- In patients with hyperglycemia (high blood sugar levels), intravenous administration of oxytocin in doses of 0.1 IU/kg lowered blood sugar levels. [215]
- In patients with type 1 diabetes mellitus, oxytocin administration at 0.2 mU/min over 60 min enhanced blood sugar recovery from hypoglycemia (low blood sugar levels). [216]
- In diabetic rats, oxytocin treatment via infusion showed histological regenerative changes of pancreatic islet cells which are involved in the production of insulin. [217-218]
- In diabetic patients with obesity, oxytocin administration for 8 weeks reduced the magnitude of blood sugar intolerance by improving insulin secretion. [219]
- Studies found that oxytocin helps achieve blood sugar balance (homeostasis) by increasing glucagon (a hormone that increases blood sugar levels) during hypoglycemic episodes and increasing insulin secretion during hyperglycemic episodes. [220-223]
M. Improves Sleep Quality
Oxytocin is also a sleep-promoting hormone. This is because it has calming, anti-anxiety, and antidepressant effects that can help induce sleep. In addition, oxytocin improves sleep quality by counteracting the effects of the stress hormone cortisol. [224]
The beneficial effects of oxytocin on sleep quality are supported by a number of studies:
- In patients with obstructive sleep apnea, a disorder caused by airway blockage during sleep, intranasal oxytocin administration (40 IU) restored cardiorespiratory homeostasis and reduced breathing problems. [225-227]
- In male rats, the administration of oxytocin in the brain improved sleep-wake behavior. [228]
- In women with high oxytocin levels, an improvement in sleep quality was observed compared to women with low oxytocin levels. [229]
- In couples, intranasal administration of oxytocin enhanced closeness, interaction, and sleep quality. [230]
- An impairment in the oxytocin gene was found to be associated with poor sleep quality. [231]
Oxytocin Side Effects
Oxytocin side effects are very uncommon. There have been some side effects associated with the use of this drug wherein the patient had one of the issues listed below at some point while being on oxytocin. However, these side effects weren’t confirmed to be associated with the treatment and could have been a coincidence and not related to the use of oxytocin. Despite this, it was listed as a side effect associated with oxytocin even though these associated side effects are very uncommon.
Side effects associated with oxytocin may include the following:
- Blurred vision
- Confusion
- Increased blood pressure
- Loss of appetite
- Nausea
- Stomach pain
- Vomiting
Oxytocin Supplement
Oxytocin, often called the “love hormone” or “bonding hormone,” is a naturally occurring hormone in the body that plays a significant role in social bonding, childbirth, and emotional connections. Oxytocin levels increase during positive social interactions, touch, and even during activities that foster trust and empathy, such as group-based activities. Given its crucial role in promoting feelings of trust and connection, synthetic oxytocin supplements have gained interest for potential therapeutic uses, particularly for those with social anxiety or mood disorders.
Oxytocin supplements, usually administered via a nasal spray, are being studied for their effects on various psychological and social disorders, including autism, social anxiety, and depression. Research has shown that oxytocin may enhance social interaction and emotional recognition in people with autism spectrum disorder (ASD), where these skills are often challenging. In addition, some studies suggest that it may help alleviate symptoms of social anxiety by promoting feelings of calm and reducing social inhibition, although more extensive studies are needed to confirm these effects and determine long-term safety.
However, the use of oxytocin supplements also raises some concerns. Since oxytocin affects social bonding and emotions, altering natural oxytocin levels through supplements could lead to unintended side effects, like impairing social cognition or increasing trust too readily, potentially making individuals more vulnerable in certain social situations. Furthermore, the effects of oxytocin can vary based on individual differences and context, which complicates its therapeutic use. As a result, while oxytocin supplements hold potential for treating certain conditions, they are currently used mostly in research settings, and experts caution that more studies are needed to fully understand the risks and benefits.
Oxytocin vs Pitocin
Oxytocin and Pitocin are closely related but serve different roles in both natural physiology and medical practice. Oxytocin is a hormone naturally produced in the hypothalamus and secreted by the pituitary gland. It plays a crucial role in social bonding, sexual reproduction, and childbirth, often called the “love hormone” because it fosters connection and trust. In labor, oxytocin helps stimulate uterine contractions, facilitating childbirth, and afterward, it aids in milk ejection during breastfeeding.
Pitocin, on the other hand, is a synthetic form of oxytocin often administered in a medical setting to induce or augment labor. It mimics the effects of natural oxytocin, but because it is delivered intravenously, it can be controlled and measured to manage labor progress more precisely. Pitocin is commonly used when labor doesn’t begin on its own or when it stalls. However, it can sometimes lead to stronger, more frequent contractions, which may increase the risk of complications like fetal distress.
While both substances influence labor contractions, their effects on the body differ in important ways. Naturally produced oxytocin is released in pulses, which provides a rhythmic, gentler influence on uterine contractions. Pitocin, being administered in a continuous stream, can create more intense and sometimes painful contractions, which may increase the need for additional medical interventions. Because of these differences, many healthcare providers weigh the potential risks and benefits carefully before using Pitocin to induce labor.
How to Increase Oxytocin
Oxytocin, often called the “love hormone,” plays a crucial role in social bonding, trust, and emotional well-being. To naturally increase oxytocin, engaging in social interactions is one of the most effective methods. Activities like hugging, holding hands, or even just spending quality time with loved ones can trigger its release. Simple acts of kindness, such as complimenting someone or helping a friend, can also help elevate oxytocin levels, promoting feelings of closeness and connection.
Physical touch and sensory experiences are powerful triggers for oxytocin release. Physical intimacy, like cuddling or massage, and even petting animals have been shown to increase oxytocin levels, which is why many find comfort in being close to pets or loved ones. Additionally, activities like meditation, yoga, and deep breathing can also stimulate oxytocin indirectly by reducing stress, creating a sense of calm that allows oxytocin to be more readily produced.
Another effective approach is through activities that foster self-compassion and empathy. Practicing gratitude, whether by journaling or reflecting on positive experiences, can boost oxytocin by creating a mindset focused on warmth and appreciation. Engaging in group activities, like singing in a choir or participating in community service, fosters a sense of togetherness that stimulates oxytocin release. By prioritizing meaningful connections and a positive environment, oxytocin levels can increase, enhancing both emotional health and resilience.
Oxytocin in Labour
Oxytocin, often referred to as the “love hormone,” plays a critical role in labor and childbirth by stimulating uterine contractions. Produced naturally in the brain by the hypothalamus and released by the pituitary gland, oxytocin increases toward the end of pregnancy, signaling the body to begin the labor process. Its action on the uterine muscles promotes the rhythmic contractions necessary to dilate the cervix and progress labor. This natural release is essential to initiating and maintaining the momentum of labor.
In medical settings, synthetic oxytocin, known as Pitocin, may be administered to induce or augment labor if it isn’t progressing as expected. By mimicking the body’s natural oxytocin, Pitocin enhances the strength and frequency of contractions, helping to move labor along. However, while effective, synthetic oxytocin may sometimes lead to very strong contractions, which can cause discomfort or necessitate closer monitoring for the safety of both the mother and baby.
Oxytocin also has a calming and bonding effect, aiding mothers in managing the physical and emotional aspects of labor. After childbirth, oxytocin levels remain high, promoting bonding between mother and newborn, especially during breastfeeding. This hormone’s release during labor and afterward not only helps mothers cope with labor but also facilitates a strong emotional connection with their babies, a crucial factor in early childhood development.
Oxytocin Nasal Spray
Oxytocin nasal spray is a therapeutic form of the hormone oxytocin, designed for easy and direct administration through the nasal passages. This method allows the hormone to enter the bloodstream and brain more rapidly, bypassing the digestive system and reducing the need for high doses. Nasal delivery can be particularly effective because it enables oxytocin to reach areas in the brain associated with social behavior, emotional processing, and stress response, thereby maximizing its benefits.
Research on oxytocin nasal sprays suggests that they may have a variety of therapeutic applications. Oxytocin is often referred to as the “bonding hormone” or “love hormone” due to its natural role in enhancing social bonds, empathy, and trust. Studies have explored its potential for improving social interactions in individuals with autism spectrum disorder, reducing anxiety and stress levels, and even supporting mood stabilization in people with depression. Its impact on stress and social behavior makes it a promising adjunct in mental health and neurological therapies.
Apart from its effects on social behaviors, oxytocin nasal spray may also have physical health benefits. For instance, it has shown promise in aiding weight loss by reducing food cravings and enhancing fat metabolism. Additionally, oxytocin can improve blood sugar control by increasing insulin sensitivity, which is beneficial for individuals with diabetes or metabolic disorders. This versatile hormone therapy continues to be researched for a wide array of potential uses, with nasal spray administration offering a non-invasive and practical route for therapeutic benefits.


























 Amlexanox Infographic
Amlexanox Infographic