Potential Health Benefits of Cardarine
Cardarine benefits include enhanced endurance, improved fat metabolism, and increased energy efficiency by activating the PPAR-delta pathway. It may also support cardiovascular health and aid in reducing inflammation, making it popular among athletes and individuals seeking metabolic improvements.
- Promotes fat loss [1-9]
- Increases muscle mass and strength [10-16]
- Improves exercise endurance [14, 17-18]
- Lowers cholesterol levels [4, 19-24]
- Improves brain health [25-29]
- Lowers risk of heart disease [1, 30-34]
- Improves blood sugar levels [14, 35-39]
- Fights kidney disease [40-43]
- Improves liver health [39, 44-51]
- Prevents and Treats Cancer [52-54]
Key Takeaways
- Enhances Endurance – Cardarine activates the PPAR-delta pathway, improving stamina and overall physical performance.
- Boosts Fat Metabolism – It promotes fat oxidation, helping the body burn stored fat more efficiently.
- Supports Cardiovascular Health – Research suggests it may improve heart health by reducing inflammation and enhancing blood vessel function.
- Increases Energy Efficiency – By optimizing mitochondrial activity, Cardarine helps the body use energy more effectively.
- Not a Steroid – Unlike anabolic steroids, Cardarine does not affect hormone levels, making it a distinct performance-enhancing compound.
What is Cardarine (GW-501516)?
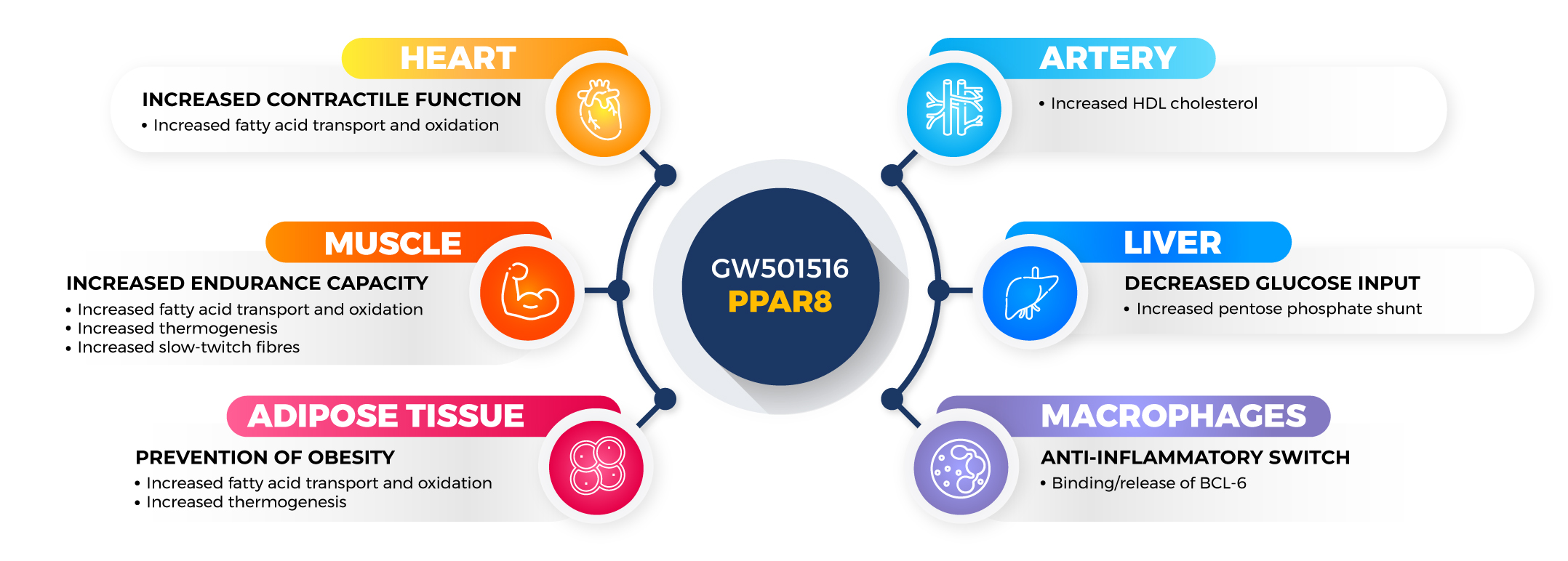
Cardarine, also known by GW501516, was initially prescribed for the treatment of various disorders related to elevated cholesterol levels such as atherosclerosis, myocardial infarction, stroke, and other blood vessel diseases. Today, this drug has gained popularity among athletes and bodybuilders due to its ability to improve muscle strength and exercise endurance. Researchers believe that cardarine exerts its beneficial effects by activating the peroxisome proliferator-activated receptor-delta (PPAR-delta) pathway. Activation of the PPAR-delta pathway is associated with increased energy levels, fat reduction, muscle building, increased endurance, and decreased blood levels of cholesterol.
How does Cardarine work?
Cardarine works by increasing muscle cell metabolism and decreasing fat deposits by stimulating lipolysis or fat breakdown. Its active ingredient can significantly increase muscle growth and endurance. Cardarine also helps regulate cholesterol levels and maintain liver health. All of these beneficial effects can be attributed to cardarine’s ability to activate the peroxisome proliferator-activated receptor-delta (PPAR-delta) pathway.
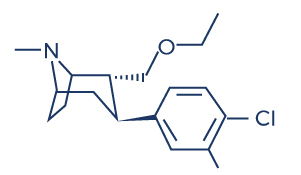
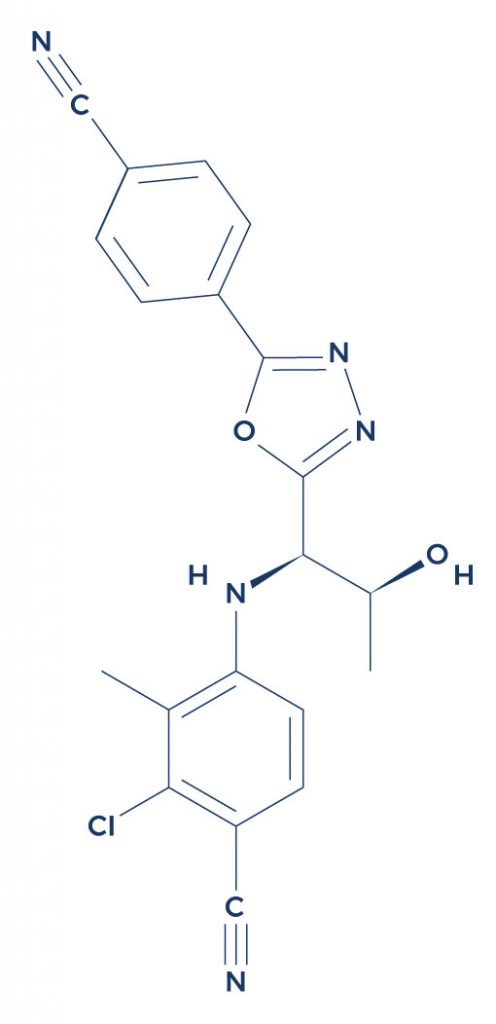
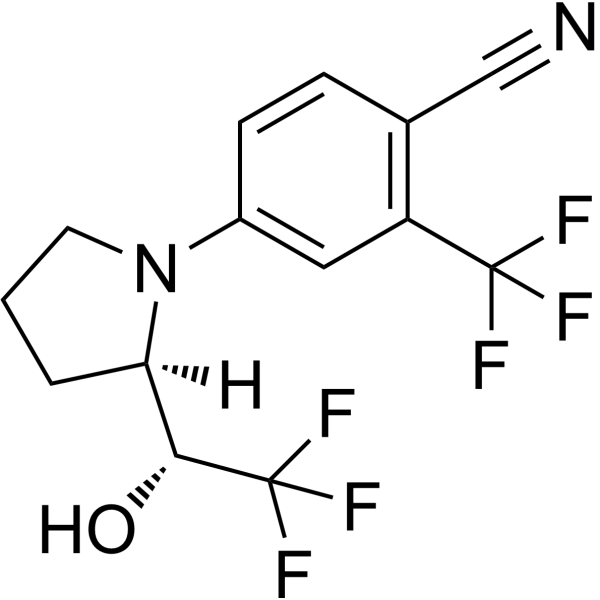
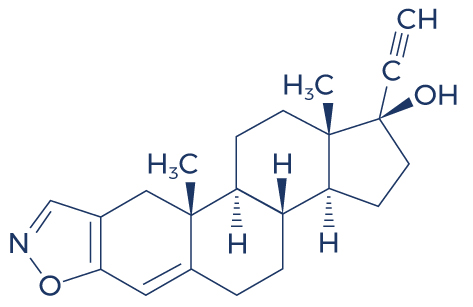
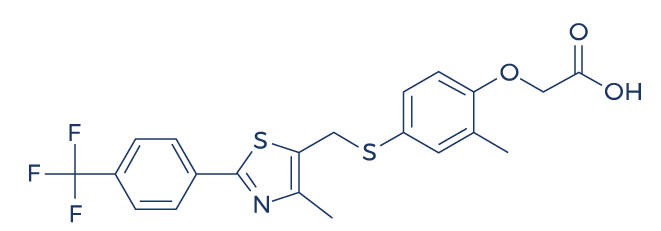
Chemical Structure of Cardarine
Research on Cardarine
Promotes Fat Loss

Cardarine (GW501516) is a compound that has gained popularity for its potential to promote fat loss, primarily by enhancing fat oxidation and increasing endurance during physical activity. It works by activating the peroxisome proliferator-activated receptor delta (PPARδ), which leads to increased mitochondrial activity, boosting the body’s ability to burn fat for energy rather than storing it. This can result in improved exercise performance, greater energy expenditure, and more efficient fat utilization, making it a potential aid in weight management and body composition improvement. However, it’s important to note that Cardarine’s use is controversial due to safety concerns and its ban by major sporting organizations.
- In men with high belly fat, administration of 2.5 mg of cardarine daily for 6 weeks resulted in weight reduction. [1]
- In inactive volunteers, subjects treated with cardarine burned more fats (20%) compared to untreated subjects. [2]
- In mice, cardarine protected against diet-induced obesity. [3]
- In moderately obese men, cardarine reduced weight by increasing fatty acid oxidation. [4]
- In overweight and obese men and women, cardarine reduced body weight by improving cholesterol profile. [5]
- A study found that cardarine can significantly reduce weight by correcting the causes of insulin resistance and abnormal cholesterol profile. [6]
- A study also found that cardarine helps reduce weight by increasing energy expenditure in muscles. [7]
- In overweight and obese healthy volunteers, cardarine treatment at a dose of 10 mg for 12 weeks reduced body fat levels. [8]
- A cell study found that cardarine activates pathways involved in fat metabolism. [9]
Increases Muscle Mass and Strength

Cardarine, also known as GW-501516, is often touted for its potential to enhance endurance and fat burning, but some claim it can indirectly promote muscle mass and strength by improving exercise performance. By enhancing the body’s ability to burn fat for fuel, Cardarine can help athletes maintain lean body mass during training, allowing them to focus more on strength-building exercises. Its role in improving cardiovascular endurance and stamina enables individuals to engage in longer, more intense workouts, which, over time, can lead to increased muscle mass and enhanced strength. However, its direct effects on muscle growth are not as pronounced as those of anabolic agents or other performance enhancers.
- Studies show that cardarine improves muscle health by regulating many different biological activities such as skeletal reprogramming, mitochondrial respiration, lipid and lipoprotein metabolism, body heat production, and muscle regeneration. [10-12]
- Studies show that cardarine improves exercise-induced reprogramming of muscle fibers by regulating the formation of genes associated with contractile proteins. [13]
- A study found that trained mice treated with cardarine had 113% more muscle fibers than untrained sedentary mice. [14]
- In mice with muscle abnormalities, cardarine treatment restored the integrity of skeletal muscle fibers. [15]
- A study found that PPAR-delta activator like cardarine regulates muscle fiber contraction and metabolism. [16]
Improves Exercise Endurance
Cardarine (GW501516) is a popular compound known for its potential to enhance exercise endurance. It works by activating the PPAR-δ receptor, which in turn boosts fat metabolism and increases the body’s ability to utilize stored fat for energy, sparing glycogen during prolonged physical activity. This mechanism can lead to improved stamina, allowing for longer and more intense workouts. Many users report experiencing enhanced endurance, quicker recovery times, and greater overall performance in endurance-based activities like running, cycling, and swimming. However, it’s important to note that the long-term safety and efficacy of Cardarine are still subjects of ongoing research.
- In both trained and untrained mice, cardarine treatment enhanced running endurance. [14]
- In adult mice, cardarine administration along with exercise training improved running endurance. [17]
- In sedentary mice, cardarine administration improved running endurance by preserving blood sugar. [18]
Lowers Cholesterol Levels
Cardarine (GW-501516) is often associated with benefits in improving cardiovascular health, particularly by lowering cholesterol levels. Studies suggest that Cardarine may help reduce LDL (bad) cholesterol while increasing HDL (good) cholesterol, which contributes to better lipid profiles and a reduced risk of heart disease. This compound works by activating the PPAR-δ receptor, which regulates fat metabolism and enhances the oxidation of fatty acids. As a result, it can support better cholesterol regulation and improve overall cardiovascular health, although it should be used with caution and under proper supervision due to potential side effects.
- In moderately obese men, treatment with cardarine reduced low density lipoprotein cholesterol by 23%. [4]
- In subjects with abnormally low high-density lipoprotein cholesterol levels, cardarine increased the levels of good cholesterol in just 12 weeks of treatment. [19]
- In patients with abnormal cholesterol profile, cardarine treatment reduced low density lipoprotein and increased high density lipoprotein cholesterol levels. [20-21]
- In healthy volunteers, cardarine enhanced the levels of high-density lipoprotein. [22]
- Administration of cardarine in patients with abnormal cholesterol profile for 12 weeks significantly reduced low-density lipoprotein. [23]
- In insulin-resistant middle-aged obese rhesus monkeys, cardarine administration resulted in a dramatic dose-dependent rise in blood levels of high-density lipoprotein cholesterol while lowering the levels of small-dense low-density lipoprotein and fasting triglycerides. [24]
Improves Brain Health
Cardarine (GW-501516) is primarily known for its effects on endurance and fat metabolism, but there is some evidence suggesting it may also have potential benefits for brain health. It activates the PPARδ receptor, which plays a role in improving mitochondrial function and enhancing neuroprotection. This could help reduce oxidative stress and inflammation in the brain, potentially supporting cognitive function and reducing the risk of neurodegenerative conditions. However, while studies in animals have shown promise, further research is needed to fully understand its impact on human brain health and its long-term safety.
- In mice, cardarine improved cognitive function by increasing blood flow to the brain. [25]
- In rat brain cells, treatment with cardarine reduced brain inflammation. [26]
- A cell study found that cardarine can lower the risk for central nervous system disorders by modulating inflammatory signaling network in the cells of the immune system. [27]
- In mice, cardarine improved spatial memory by promoting formation of new neurons (neurogenesis). [28]
- In a model of brain inflammation, cardarine protected against nerve cell damage by reducing inflammatory processes. [29]
Lowers Risk of Heart Disease
Cardarine (GW501516) is a synthetic compound that has been shown to potentially lower the risk of heart disease by improving lipid profiles and enhancing cardiovascular health. It works by activating the peroxisome proliferator-activated receptor delta (PPARδ), which plays a crucial role in fat metabolism and energy expenditure. By increasing the oxidation of fatty acids, Cardarine helps reduce triglyceride levels and raises high-density lipoprotein (HDL) cholesterol, often referred to as “good” cholesterol. Additionally, it may reduce the accumulation of visceral fat and improve overall blood vessel function, which can contribute to a reduced risk of atherosclerosis and other heart-related issues. However, its long-term safety and efficacy require further research to fully understand its impact on heart health.
- Cardarine may reduce the risk of plaque build-up in the arteries of the heart (atherosclerosis) by relaxing/widening the blood vessels via nitric oxide production. [1]
- Cardarine prevents atherosclerosis by antagonizing multiple proinflammatory pathways. [30]
- Cardarine also has the ability to prevent the formation of lesions. [31-32]
- Cardarine stimulates the growth of new blood vessels in the heart by boosting the levels of vascular endothelial growth factor (VEGF). [33]
- Cardarine improves overall heart health by regulating cardiomyocyte (heart cell) proliferation and cardiac repair. [34]
Improves Blood Sugar Levels
Cardarine (GW501516) has been shown to improve blood sugar levels by enhancing insulin sensitivity and promoting glucose uptake in muscles. This can lead to more efficient use of glucose, preventing excess buildup in the bloodstream. It works by activating the peroxisome proliferator-activated receptor delta (PPAR-δ), which increases fatty acid oxidation and helps regulate energy metabolism. As a result, Cardarine may support better blood sugar control, particularly in individuals with insulin resistance or metabolic syndrome, although its use should be approached with caution due to concerns over long-term safety.
- In mice, cardarine enhanced specific consumption of fatty acids and reduced blood sugar utilization. [14]
- In high fat-fed rats and mice, cardarine improved insulin response which in turn reduced blood sugar levels. [35]
- In a mouse model of metabolic syndrome, cardarine administration ameliorated insulin resistance. [36]
- In mice fed with a high-fat diet, cardarine improved blood sugar levels by enhancing insulin signaling. [37]
- Cardarine also improved blood sugar in mice with obesity-related disorders by regulating blood sugar metabolism and insulin sensitivity. [38]
- In mice, cardarine prevented cytokine-induced insulin resistance in liver cells. [39]
Fights Kidney Disease
Cardarine (GW501516) is a compound often linked to performance enhancement, but some studies suggest it may also offer benefits in fighting kidney disease. It is believed to work by activating the PPARδ (peroxisome proliferator-activated receptor delta), which helps regulate fat metabolism, reduce inflammation, and improve mitochondrial function. These actions may support kidney health by reducing oxidative stress, lowering inflammatory markers, and improving overall cellular function within kidney tissues. However, while these potential benefits are promising, more clinical research is needed to conclusively determine its effectiveness in combating kidney disease and its long-term safety.
- Cardarine protects against kidney disease by reducing the activity of MCP-1, a gene related to kidney disorders. [40]
- In high-fructose fed mouse model, cardarine improved inflammatory pathways in the kidneys. [41-42]
- A study found that cardarine has the potential to reduce kidney inflammation, thus, preventing kidney disease progression. [43]
Improves Liver Health
Cardarine (GW501516) has been suggested to improve liver health by promoting fat metabolism and reducing inflammation in the liver. It works by activating the peroxisome proliferator-activated receptor delta (PPARδ), which enhances the body’s ability to burn fat for energy, potentially reducing the accumulation of fat in liver cells, a key contributor to fatty liver disease. Additionally, Cardarine may help reduce oxidative stress, which can damage liver cells, leading to better overall liver function. However, it’s important to note that while some animal studies show promising results, further research is needed to fully understand its long-term effects on human liver health.
- A study found that cardarine has the potential to treat kidney diseases associated with metabolic syndrome. [44]
- In animal models of nonalcoholic fatty liver disease, cardarine treatment reduced liver inflammation. [45]
- In mice, cardarine treatment significantly reduced the prevalence of liver damage from a high-fructose diet and the development of nonalcoholic fatty liver disease. [39, 46]
- In animal models of non-alcholic steatohepatitis (fat build-up in the liver), cardarine ameliorated symptoms by enhancing fatty acid β-oxidation. [47-50]
- A study found that cardarine and other peroxisome proliferator-activated receptors have the ability to stimulate liver regeneration by modulating Akt and E2f Signaling. [51]
Prevents and Treats Cancer
Cardarine (GW501516) is a synthetic compound that has shown promise in research for its potential to prevent and treat cancer. It works by activating the PPARδ (peroxisome proliferator-activated receptor delta) pathway, which plays a key role in regulating metabolism, inflammation, and cellular growth. Studies suggest that Cardarine can help inhibit the proliferation of cancer cells and reduce tumor growth, particularly in cancers like breast, colon, and liver. Its anti-inflammatory effects, combined with its ability to enhance fat metabolism, may contribute to its potential as a cancer prevention and treatment agent. However, it’s important to note that more clinical trials are needed to fully understand its efficacy and safety in humans.
- A study showed the anti-inflammatory properties of cardarine in human pancreatic cancer. [52]
- In mice, cardarine inhibited tumor growth in nasopharyngeal carcinoma (NPC). [53]
- A study showed that cardarine induced apoptosis in invasive bladder cancer cells. [54]
Cardarine Side Effects
Cardarine side effects are very uncommon. There have been some side effects associated with the use of this drug wherein the patient had one of the issues listed below at some point while being on cardarine. However, the issue wasn’t’ confirmed to be caused by the treatment and could have been a coincidence and not related to the use of cardarine. Despite this, it was listed as a side effect associated with cardarine even these associated side effects are very uncommon.
Side effects associated with cardarine may include the following:
- Dizziness
- Flushing
- Headache
- Lightheadedness
- Swelling of the ankles/feet
Cardarine vs ostarine
Cardarine, Ostarine, and MK-677 are all popular compounds in the fitness and bodybuilding community, but they serve different purposes. Cardarine (GW-501516) is a PPAR-delta agonist designed to enhance endurance, boost fat metabolism, and improve cardiovascular health. It does not affect muscle growth directly but helps users sustain longer workouts and burn fat more efficiently.
Ostarine (MK-2866), on the other hand, is a selective androgen receptor modulator (SARM) primarily used for muscle preservation and growth. It helps prevent muscle loss during cutting phases and supports lean muscle gains without the androgenic side effects of steroids. Unlike Cardarine, Ostarine directly influences muscle tissue by binding to androgen receptors, promoting muscle repair and growth. MK-677, also known for its growth hormone-releasing effects, can be stacked with these compounds to promote further muscle recovery and enhance muscle mass.
While both Cardarine and Ostarine can be used together in a cutting cycle, and MK-677 can support the process by improving muscle repair, they have distinct mechanisms of action. Cardarine focuses on endurance and fat oxidation, while Ostarine helps maintain muscle mass, and MK-677 supports muscle growth and recovery. Choosing between them depends on individual fitness goals—Cardarine is ideal for enhancing stamina and fat loss, while Ostarine is better suited for muscle retention and recovery, with MK-677 complementing these effects for overall muscle development.
Cardarine cycle length
Cardarine, Ostarine, and MK 677 are all popular compounds in the fitness and bodybuilding community, but they serve different purposes. Cardarine (GW-501516) is a PPAR-delta agonist designed to enhance endurance, boost fat metabolism, and improve cardiovascular health. It does not affect muscle growth directly but helps users sustain longer workouts and burn fat more efficiently.
Ostarine (MK-2866), on the other hand, is a selective androgen receptor modulator (SARM) primarily used for muscle preservation and growth. It helps prevent muscle loss during cutting phases and supports lean muscle gains without the androgenic side effects of steroids. MK 677, another compound used for muscle growth, promotes the secretion of growth hormone, further enhancing the potential for muscle recovery and growth. Unlike Cardarine, Ostarine and MK 677 directly influence muscle tissue by binding to androgen receptors and stimulating growth hormone release, respectively.
While all three compounds can be used together in a cutting cycle, they have distinct mechanisms of action. Cardarine focuses on endurance and fat oxidation, while Ostarine and MK 677 help maintain muscle mass and promote recovery. Choosing between them depends on individual fitness goals—Cardarine is ideal for enhancing stamina and fat loss, while Ostarine and MK 677 are better suited for muscle retention and growth.
Cardarine capsules
Cardarine capsules are a popular oral form of the PPAR-delta agonist, designed for convenient dosing and optimal absorption. These capsules are commonly used by athletes and fitness enthusiasts seeking to enhance endurance, boost fat metabolism, and improve energy efficiency without affecting hormone levels. Their standardized dosage makes it easier to track intake and maintain consistent results.
When taken as directed, Cardarine capsules work by stimulating the PPAR-delta pathway, increasing the body’s ability to burn fat while preserving muscle mass. This makes them especially appealing for individuals looking to improve body composition and athletic performance. Additionally, some research suggests potential cardiovascular benefits, such as reducing inflammation and improving lipid profiles.
Despite their potential advantages, users should be aware of ongoing debates regarding the long-term safety of Cardarine. While animal studies have raised concerns about prolonged use, human research is still limited. As with any supplement, it’s essential to source Cardarine capsules from reputable suppliers and consult a healthcare professional before use.
Cardarine and alcohol
Cardarine blood work is essential for monitoring its effects on lipid profiles, liver function, and overall metabolic health. Studies suggest that Cardarine can significantly improve cholesterol levels by increasing HDL (good cholesterol) and reducing LDL (bad cholesterol), which may contribute to cardiovascular benefits. Regular blood tests can help track these changes and ensure that lipid levels remain within a healthy range.
Liver function tests are also important when using Cardarine, as any compound affecting metabolism may influence liver enzymes. While research has not shown direct liver toxicity, routine blood work can help detect any potential abnormalities early. Checking markers such as ALT and AST levels can provide insights into liver health and ensure safe use over time.
Additionally, blood work can assess inflammation markers and glucose metabolism, as Cardarine is known to enhance insulin sensitivity. Monitoring fasting blood sugar and C-reactive protein (CRP) levels can help determine whether Cardarine is positively influencing metabolic health. Regular testing ensures that any unexpected side effects are detected early, allowing for adjustments if needed.
Cardarine blood work
Cardarine blood work is essential for monitoring its effects on lipid profiles, liver function, and overall metabolic health. Studies suggest that Cardarine can significantly improve cholesterol levels by increasing HDL (good cholesterol) and reducing LDL (bad cholesterol), which may contribute to cardiovascular benefits. Regular blood tests can help track these changes and ensure that lipid levels remain within a healthy range.
Liver function tests are also important when using Cardarine, as any compound affecting metabolism may influence liver enzymes. While research has not shown direct liver toxicity, routine blood work can help detect any potential abnormalities early. Checking markers such as ALT and AST levels can provide insights into liver health and ensure safe use over time.
Additionally, blood work can assess inflammation markers and glucose metabolism, as Cardarine is known to enhance insulin sensitivity. Monitoring fasting blood sugar and C-reactive protein (CRP) levels can help determine whether Cardarine is positively influencing metabolic health. Regular testing ensures that any unexpected side effects are detected early, allowing for adjustments if needed.
Cardarine rad140 stack
Stacking Cardarine (GW-501516) and RAD-140 (Testolone) is popular among athletes and bodybuilders for its potential synergy in enhancing endurance, fat loss, and muscle growth. Cardarine is known for improving stamina and metabolic efficiency by activating the PPAR-delta pathway, while RAD-140, a selective androgen receptor modulator (SARM), promotes lean muscle gains and strength. Together, they offer a combination of endurance enhancement and muscle-building effects.
This stack is often used during cutting cycles, as Cardarine helps with fat oxidation while RAD-140 preserves muscle mass and boosts recovery. Users report increased workout intensity, improved vascularity, and a leaner physique. Additionally, since Cardarine does not suppress natural testosterone production, it may help counterbalance some of the hormonal suppression caused by RAD-140.
Despite the benefits, potential side effects should be considered. RAD-140 can suppress testosterone, requiring post-cycle therapy (PCT), while Cardarine’s long-term safety remains debated due to animal studies linking it to cancer risk. Careful dosing, cycle length management, and regular health monitoring are essential for minimizing risks and maximizing results from this stack.
How long does cardarine take to work
Cardarine typically begins to take effect within a few days to a week, with users reporting increased endurance and energy levels early on. Since it activates the PPAR-delta pathway, which enhances fat metabolism and muscle efficiency, some benefits may be noticeable relatively quickly, especially during physical activity. However, the full effects on fat loss and endurance improvement may take a few weeks to become more pronounced.
Most users experience significant results within 4 to 6 weeks of consistent use, particularly in terms of stamina and fat oxidation. As the body adapts to the increased mitochondrial activity and improved metabolic efficiency, endurance continues to improve over time. Individual response varies based on dosage, activity level, and overall lifestyle factors like diet and exercise.
By the 8 to 12-week mark, users often see peak benefits, including noticeable improvements in cardiovascular performance and body composition. However, as with any performance-enhancing compound, results depend on sustained use, and cycling protocols are often recommended to optimize long-term effectiveness while minimizing potential risks.
Cardarine supplement
Cardarine is a research chemical often marketed as a supplement for its potential to enhance endurance, boost fat metabolism, and improve energy efficiency. Originally developed for treating metabolic and cardiovascular disorders, it gained popularity among athletes and fitness enthusiasts due to its ability to activate the PPAR-delta pathway, which promotes fat oxidation and muscle endurance. Unlike traditional stimulants, Cardarine does not act on the central nervous system, making it appealing for those looking to improve performance without jittery side effects.
Despite its promising benefits, Cardarine remains an experimental compound, and its long-term safety profile is not fully understood. Some studies in animal models raised concerns about potential cancer risks with prolonged use, leading to its discontinuation in pharmaceutical research. As a result, it is not approved for human consumption by regulatory bodies like the FDA and is primarily sold as a research chemical rather than a dietary supplement.
Individuals considering Cardarine should be cautious and aware of potential risks, as well as the legal status in their region. While some users report positive effects on endurance and fat loss, the lack of clinical trials in humans means its safety and efficacy remain uncertain. Consulting a healthcare professional before use is essential, especially for those with underlying health conditions or those subject to drug testing regulations.
Cardarine for women
Cardarine is often used by women seeking to improve endurance, boost fat metabolism, and enhance overall athletic performance. Unlike anabolic steroids, it does not interfere with hormone levels, making it an appealing option for women looking to increase stamina without the risk of masculinizing side effects. Its ability to promote fat oxidation can also help with body composition goals, making it a popular choice among female athletes and fitness enthusiasts.
For women, the typical Cardarine dosage is lower than that used by men, often ranging from 5 to 10 mg per day. This helps minimize any potential side effects while still providing significant benefits in endurance and fat burning. Since Cardarine does not directly stimulate muscle growth like steroids, it is commonly stacked with other supplements or training programs focused on lean muscle development.
While Cardarine is not known to cause hormonal imbalances, long-term safety remains a concern due to limited human research. Women considering its use should be cautious, monitor their response closely, and consult with a healthcare professional before starting. Additionally, sourcing high-quality products is essential to avoid contamination or counterfeit substances, as Cardarine is often sold in the research chemical market.
Cardarine dosage for males
The optimal Cardarine dosage for males typically ranges from 10-20 mg per day, depending on individual goals and experience with the compound. Beginners often start with 10 mg daily to assess tolerance, while more experienced users may increase to 20 mg per day for enhanced endurance and fat metabolism. It is usually taken once daily due to its long half-life of around 24 hours.
A common cycle length for Cardarine is 6-12 weeks, followed by a break to allow the body to reset. While it does not suppress natural testosterone production, taking periodic breaks can help minimize potential risks. Some users choose to stack Cardarine with other compounds like SARMs for synergistic benefits, but caution is advised when combining substances.
Since Cardarine is not liver-toxic, it does not require post-cycle therapy (PCT). However, maintaining a healthy diet and exercise routine is essential to maximize its effects. As with any research compound, consulting a healthcare professional before use is recommended to ensure safety and proper dosing.
When to take cardarine
The timing of taking Cardarine largely depends on your personal fitness goals and routine. For those using Cardarine to enhance endurance and performance, it’s commonly recommended to take it about 30 to 60 minutes before a workout. This timing allows the compound to activate the PPAR-delta pathway, optimizing fat metabolism and energy utilization during exercise, leading to better performance and stamina.
If you’re using Cardarine for fat loss or metabolic support, it can be taken at any time during the day, as it works by improving overall fat oxidation and energy efficiency. Some prefer to take it in the morning with their first meal to start the day with an energy boost, while others take it before a workout for maximum benefit during physical activity.
As with any supplement, consistency is key. To maintain stable levels in your system, taking Cardarine at the same time each day, whether before exercise or at a time that suits your routine, can help you achieve the best results over time. However, it’s essential to follow the recommended dosage and consult with a healthcare provider, particularly if combining it with other supplements or medications.
Cardarine dosage
Cardarine dosage typically starts at a lower range to assess individual tolerance and gradually increases if necessary. A common starting dose for athletes and individuals using Cardarine for performance enhancement is around 10 mg per day. This dosage is often taken once daily, although some users may split the dose into two smaller servings to reduce potential side effects. It’s important to note that the optimal dosage can vary based on factors like body weight, goals, and response to the compound.
For those seeking to maximize endurance or fat-burning effects, higher doses of up to 20 mg per day may be used, but it is essential to stay within safe limits to minimize any risks. Doses exceeding 20 mg per day are generally not recommended, as the long-term safety of higher doses is not well-established. Consistent use over extended periods is not typically advised, as there is limited data on the long-term effects of Cardarine.
As with any supplement or performance-enhancing compound, it is crucial to consult with a healthcare professional before starting Cardarine, especially if there are underlying health conditions. Monitoring for any adverse effects or unusual symptoms is recommended, and users should cycle off the compound after a set period to give the body a break and reduce the risk of potential side effects.
Ostarine and cardarine stack
The combination of Ostarine (MK-2866) and Cardarine (GW-501516), often referred to as a “stack,” is popular among athletes and fitness enthusiasts looking to improve performance, enhance fat loss, and increase endurance. Ostarine is a selective androgen receptor modulator (SARM) known for promoting lean muscle mass and strength without the side effects commonly associated with anabolic steroids. Cardarine, on the other hand, is a PPAR-delta activator that enhances endurance, fat metabolism, and cardiovascular health. Together, they create a synergy that can accelerate fat loss while maintaining or even increasing lean muscle mass.
Ostarine works by binding to androgen receptors in muscle tissue, stimulating muscle growth and repair, while Cardarine helps to improve the body’s ability to burn fat by boosting metabolic efficiency and increasing endurance. This combination makes the stack appealing for individuals aiming to get leaner, more toned, and stronger, as it helps maximize fat loss while preserving muscle. Moreover, Cardarine’s endurance-enhancing effects allow users to push harder in training, potentially leading to more effective workouts and greater overall progress.
However, while the Ostarine and Cardarine stack can offer performance benefits, it is important to approach it with caution, as both substances are not approved for long-term use outside of research settings. Side effects such as hormone imbalances with Ostarine or potential liver strain with Cardarine are concerns to monitor. Always consider consulting with a healthcare professional before using such compounds, especially when combining them for enhanced performance.
Ostarine and cardarine
Ostarine and Cardarine are both popular compounds in the fitness and bodybuilding communities, though they serve different purposes. Ostarine, also known as MK-2866, is a selective androgen receptor modulator (SARM) that promotes muscle growth, strength, and fat loss by binding to androgen receptors in the body. It is often used by athletes looking to enhance muscle mass without the negative side effects of anabolic steroids. Ostarine can help improve recovery, increase lean muscle, and support fat reduction during cutting phases.
Cardarine, on the other hand, is not a SARM but a PPAR-delta agonist. It works by activating the PPAR-delta pathway, which helps increase endurance, improve fat metabolism, and enhance cardiovascular performance. Athletes often use Cardarine to boost stamina and energy efficiency, allowing them to train harder and longer. Its fat-burning properties also make it a popular choice for those looking to improve body composition and overall metabolic health.
While both Ostarine and Cardarine are used to enhance athletic performance, they work through different mechanisms in the body. Ostarine primarily supports muscle growth and recovery, while Cardarine is more focused on increasing endurance and fat oxidation. Combining the two can provide synergistic benefits, as users may experience muscle gain and improved endurance, making them a popular pairing in training regimens. However, it is important to note that the long-term safety and regulatory status of these compounds remain uncertain, and they are not approved by major sports organizations for competition.
What does cardarine do
Cardarine, also known as GW-501516, is a synthetic compound that activates the PPAR-delta pathway, a receptor involved in regulating metabolism. By activating this pathway, Cardarine increases the body’s ability to oxidize fat, improving fat-burning processes and enhancing endurance. It is often used by athletes and bodybuilders to boost stamina during prolonged physical activity, as it helps the body use fat as a primary energy source rather than carbohydrates.
In addition to boosting endurance and fat metabolism, Cardarine is believed to support cardiovascular health. Research suggests that it can reduce inflammation and improve blood vessel function, contributing to better overall heart health. This makes it potentially beneficial for individuals looking to improve their cardiovascular fitness and reduce the risk of metabolic diseases such as obesity or diabetes.
While Cardarine does not influence hormones like anabolic steroids, it is valued for its ability to enhance energy efficiency and physical performance. Its effects on fat metabolism and endurance make it a popular choice for those seeking to improve exercise output and body composition, though long-term safety and regulatory status require further study.
Stenabolic vs cardarine
Stenabolic and Cardarine are both performance-enhancing compounds that target similar pathways in the body, but they differ in their mechanisms and uses. Stenabolic, also known as SR9009, is a Rev-Erb agonist that regulates circadian rhythms and boosts metabolism, leading to improved endurance, fat loss, and energy expenditure. It is often favored by athletes looking for enhanced physical performance and fat-burning effects without the need for stimulants.
Cardarine, also known as GW501516, works by activating the PPAR-delta pathway, which increases fat oxidation and improves endurance. It is primarily used by individuals aiming to improve cardiovascular health, increase stamina, and enhance fat metabolism. Cardarine is well-known for its ability to help burn fat while maintaining muscle mass, making it a popular choice for those focused on weight management and athletic performance.
While both compounds offer similar benefits, such as improved endurance and fat loss, their safety profiles and potential long-term effects differ. Stenabolic has not been extensively studied in humans, and there are concerns about its safety, particularly related to potential liver toxicity. On the other hand, Cardarine was linked to cancer development in animal studies, leading to concerns about its long-term use. Both substances should be approached with caution, and users should carefully consider the risks before incorporating them into their regimen.